By Vikrant Rana and Huda Jafri
In the pharmaceutical and biotechnology industries, a molecule is the starting point – but a brand is the destination. From a patented active ingredient to a household name, the journey from laboratory to patient involves one of the most legally intricate and commercially significant areas of intellectual property law: pharmaceutical trademark strategy.
India stands at the heart of this story. As the world’s third-largest trademark filing jurisdiction and the supplier of 20% of global generic medicines by volume, India’s pharmaceutical sector commands both domestic and global attention. At S.S. Rana & Co., we advise innovator pharma companies, generic manufacturers, biotech startups, and multinational corporations on every aspect of pharmaceutical IP – from brand clearance to enforcement in the Supreme Court of India.
This article distils the key principles of pharmaceutical and biotechnology trademark law, grounded in current market data, Indian legislation, and landmark case law.
1. India’s Pharmaceutical Sector: The Market Context
India’s pharmaceutical industry has emerged as a global powerhouse. As of 2024–25, the sector’s headline figures tell a compelling story:[1]
| Total Market Size | US$ 50 Billion+ |
| Domestic Consumption | US$ 23.5 Billion |
| Pharmaceutical Exports | US$ 26.5 Billion |
| Share of Global Generics | 20% by volume |
| Manufacturing Scale | 10,500+ manufacturing units; 3,000+ pharma companies |
| Export Reach | 200+ countries |
| Projected Size by 2030 | US$ 130 Billion (IBEF projection)[2] |
| Global Pharma Market (2024) | US$ 1.68 Trillion; projected US$ 3.07 Trillion by 2034 (6.2% CAGR)[3] |
2. The Counterfeiting Crisis: Why Brand Protection Is a Patient Safety Imperative
The scale of pharmaceutical counterfeiting is a global public health emergency, and it underlines why strong trademark rights are not merely commercial instruments – they are instruments of patient safety:
- 1 in 10 medicines in low- and middle-income countries are substandard or falsified, according to the WHO Global Surveillance & Monitoring System.[4]
- The estimated global market for fake pharmaceutical products is US$ 200 billion, with 67% of these products categorised as dangerous to human health.[5]
- The majority of substandard and falsified drugs are sold online or in informal markets, bypassing regulatory controls entirely.
- Fake antibiotics and sub-potent drugs are a leading driver of growing antimicrobial resistance globally.
- In India, the Drugs Controller General issued quality alerts on 53 drug samples that failed quality standards in 2024.[6]
- The proliferation of illegal online pharmacies has made border and platform enforcement a critical front in anti-counterfeiting strategy.
| A registered trademark is not merely a commercial asset – in pharma, it is a front line of consumer protection. Counterfeiting a pharmaceutical brand is counterfeiting patient safety. |
3. Why Trademarks Matter in Pharma: Six Core Benefits
Unlike patents, which expire in 20 years, a pharmaceutical trademark is renewable indefinitely. This makes it the most enduring IP asset in any pharmaceutical company’s portfolio. The specific benefits are:
- Medical Error Reduction: Distinctive marks reduce prescribing and dispensing confusion, directly protecting patient safety.
- Brand Protection: Prevents competitors from using confusingly similar brand names, logos, or trade dress.
- Consumer Choice: Enables patients and physicians to identify and choose preferred medicines with confidence.
- Product Credibility: A registered trademark signals quality and builds lasting trust with healthcare professionals.
- Counterfeit Combat: Legal exclusivity over a mark creates the enforcement foundation needed to pursue counterfeiters.
- Enduring Commercial Value: Brand equity survives patent expiry. When Crocin®’s active ingredient paracetamol entered the public domain, the brand name remained GSK’s exclusive asset.
| Key Insight: Unlike a patent (20-year fixed term), a pharmaceutical trademark lasts indefinitely – renewable every 10 years, forever. It is the longest-lasting IP protection available in the pharmaceutical arsenal. |
4. What Can Be Trademarked in Pharma? A Comprehensive Scope
Section 2(1)(zb) of the Trade Marks Act, 1999 defines a trademark broadly to include any mark capable of being represented graphically and capable of distinguishing goods or services. In the pharmaceutical context, the following categories of marks are protectable:[7]
| Image | Type | Application No. | Class | TM Applied For | Proprietor Name |
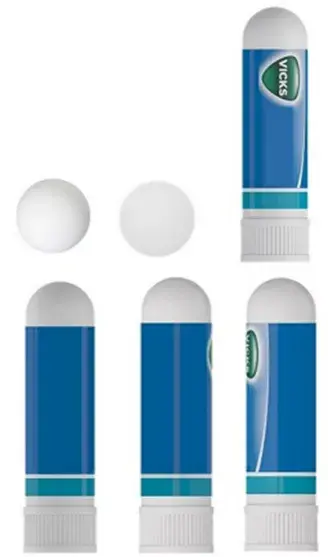 |
3D / Shape Mark | 5262628 | 5 | VICKS INHALER – 3D Mark | The Procter & Gamble Company |
 |
Logo / Device Mark | 1501137 | 99 | VICKS | The Procter & Gamble Company |
 |
Packaging / Device Mark | 2442061 | 5 | BENADRYL (Device) | Johnson & Johnson |
| Word Mark | 1592383 | 4 | MOOV | Reckitt Benckiser Healthcare India Pvt. Ltd. |
5. Trademark Prosecution: The Critical Path from Concept to Registration
5.1 Start with Clearance
Before a pharmaceutical company invests in brand development, packaging, and regulatory filing, a thorough trademark clearance search is essential. Poor pre-launch planning in pharma is extraordinarily costly because a post-launch name change triggers not just a commercial rebrand – it requires a fresh drug name approval from the Central Drugs Standard Control Organisation (CDSCO), adding months or years to the commercial timeline.
Strong marks for clearance share these characteristics: coined or invented words; phonetically unique and not similar to existing INNs; visually distinctive; not descriptive of the drug’s indication, dosage, or composition. Weak marks that are more likely to face objections include those that are descriptive of drug use, similar to INN stems, phonetically similar to existing registrations, generic or laudatory terms.
5.2 Where to Search
A thorough clearance exercise covers all of the following databases:
- Indian Trademark Registry – www.ipindia.nic.in (phonetic and exact-match search)
- WIPO Global Brand Database – www.wipo.int/branddb (for Madrid Protocol registrations)
- Ministry of Company Affairs – www.mca.gov.in (company/trade names that may block registration)
- CDSCO Drug Approvals Portal – www.cdsco.gov.in (approved drug names already in regulatory use)
- WHO INN Database – www.who.int/medicines/inn (conflicts with international nonproprietary names)
- Domain Name Availability – for digital brand consistency
5.3 Grounds for Refusal: What Cannot Be Registered
The Trade Marks Act, 1999 sets out three key categories of non-registrable marks especially relevant to pharma:
Section 9 (Absolute Grounds): Marks devoid of distinctive character; descriptive of goods or services; customary in current language; contrary to public policy; deceptive as to nature, quality, or origin.
Section 11 (Relative Grounds): Marks identical or similar to earlier registered marks; marks that would cause confusion with well-known marks; marks that take unfair advantage of a prior mark’s reputation.
Section 13 (Prohibition): Names of chemical elements, chemical compounds, and International Nonproprietary Names (INNs), or marks deceptively similar to any INN, cannot be registered as trademarks. This is a hard statutory bar.
6. The INN System: Why Generic Names Belong to Everyone
International Nonproprietary Names (INNs) are assigned by the World Health Organisation (WHO) to uniquely and unambiguously identify each pharmaceutical active substance. INNs are placed in the public domain and may be freely used by any manufacturer, regulator, or healthcare professional worldwide – they cannot be monopolised by any single company.
Section 13 of the Trade Marks Act, 1999 gives statutory force to this principle in India: no trademark registration is permissible for an INN, a chemical element name, or any mark deceptively similar to a WHO-declared INN.[8][9]
Commonly used INN stems that are in the public domain and cannot form the basis of a pharmaceutical trademark:
| INN Stem | Drug Class |
| -mab | Monoclonal antibodies |
| -vir | Antiviral drugs |
| -statin | Cholesterol-lowering drugs |
| -pril | ACE inhibitors |
| -sartan | ARBs (blood pressure) |
| -caine | Local anaesthetics[10] |
The Three-Name System in Pharmaceuticals
Every pharmaceutical product exists under three distinct names, each with a different legal status:
| Name Type | Example (Painkiller) | Ownership & Protection |
| Chemical / IUPAC Name | N-acetyl-para-aminophenol | No one. Always public. Cannot be protected. |
| Generic / INN | Paracetamol / Acetaminophen | WHO-assigned. In public domain. Cannot be trademarked under Section 13. |
| Brand / Trademark | Crocin® (GSK), Calpol® (GSK), Tylenol® (J&J) | Company exclusive. Indefinitely renewable. Fully protected even after patent expiry. |
| Commercial Significance: When a drug patent expires, competitors can manufacture the same molecule under any name. They cannot use the original brand name. This is why Crocin® remains GSK’s exclusive commercial asset even as paracetamol became freely available to all manufacturers. |
7. Branding in Biotechnology and Biosimilars: New Frontiers
7.1 Why Biotech Branding Is Different
Biologics – including insulin, monoclonal antibodies, and gene therapies – are large, complex molecules produced from living cells. Unlike small-molecule drugs, they cannot be perfectly replicated. This creates distinctive branding and naming challenges that extend beyond conventional pharmaceutical trademark strategy:
- Platform Branding: Biotech companies now brand not just individual products but entire technology platforms. Moderna’s mRNA platform, for instance, functions as an umbrella identity covering all future mRNA-based products.
- Early-Stage Branding: Biotech startups increasingly brand their R&D platforms before any product reaches the market – building investor trust, market valuation, and competitive positioning from day one.
- Evergreening Post-Patent: When a biologic’s patent expires, multiple biosimilars enter the market. Strong brand loyalty among physicians and patients sustains originator revenue.
- Regulatory & TM Dual Track: The WHO INN application and the trademark application must run in parallel. Conflicts between an INN and a proposed brand name must be resolved before regulatory market approval is granted.
7.2 FDA Biosimilar Naming Rules
The US Food and Drug Administration (FDA) requires biosimilar products to use a distinguishing four-lowercase-letter suffix after the INN to prevent inadvertent substitution in prescribing and dispensing. Selected examples:[11]
| Originator Brand | INN (Generic) | Biosimilar Brand | FDA Suffix |
| Humira® | adalimumab | Hadlima® | -hadlm |
| Humira® | adalimumab | Hyrimoz® | -aqvh |
| Enbrel® | etanercept | Erelzi® | -szzs |
| Remicade® | infliximab | Inflectra® | -dyyb |
| Neupogen® | filgrastim | Zarxio® (first US biosimilar approved, 2015) | -sndz[12] |
8. Deceptive Similarity in Pharma: Landmark Indian Case Law
Indian courts have consistently held that the standard for deceptive similarity in pharmaceutical trademarks must be more stringent than in other industries, given that prescribing and dispensing errors can have life-threatening consequences.
8.1 Cadila Healthcare Ltd. v. Cadila Pharmaceuticals Ltd.
The landmark case. The dispute arose over FALCIGO® versus FALCITAB®, both antimalarial drugs treating Plasmodium falciparum malaria. The Supreme Court laid down the governing framework for deceptive similarity in pharma:[13]
- Degree of Resemblance: All three dimensions – phonetic, visual, and conceptual – must be assessed.
- Nature of Goods: Medical goods require stricter scrutiny given the higher harm from confusion.
- Class of Consumers: Courts must account for illiterate patients and semi-educated buyers, not merely sophisticated consumers.
- Mode of Purchase: OTC drugs versus prescription drugs attract different standards.
- Surrounding Circumstances: All contextual factors affecting the likelihood of confusion must be considered.
| “The principles governing deceptive similarity require meticulous consideration especially in pharma – even minimal confusion can lead to catastrophic consequences.” – Supreme Court of India, Cadila Healthcare v. Cadila Pharmaceuticals (2001) |
8.2 M/s Abbott GmbH v. Registrar of Trademarks
BRUFEN® versus MEBUFEN: Abbott GmbH opposed the registration of MEBUFEN on the ground of deceptive similarity to their established ibuprofen brand BRUFEN®. The Court applied the phonetic test, finding that both marks ended in ‘-FEN’, shared similar consonant patterns, and that MEBUFEN structurally contained ‘-BUFEN’. Registration of MEBUFEN was refused.[14]
Key Principle: In pharmaceutical marks, even partial phonetic similarity can constitute deceptive similarity given the patient safety imperative.
8.3 Sun Pharma v. Anglo French Drugs
OXETOL® versus EXITOL: Phonetic test applied – both marks had identical vowel sounds and stress patterns when spoken. The Court granted an injunction in Sun Pharma’s favour, holding that phonetic similarity alone is sufficient to establish deceptive similarity in pharmaceutical marks. The Court gave heightened weight to patient safety concerns given that anti-epileptic drugs, if wrongly substituted, can trigger fatal seizures.[15]
8.4 Glenmark Pharmaceuticals v. Gleck Pharma
ZITA-MET versus XIGA-MET: Both marks used for anti-diabetic combination drugs containing metformin. The Court held the ‘-MET’ suffix common to both as non-distinctive, focusing analysis on the distinctive prefix – ZITA versus XIGA. Holistic impression found to be deceptively similar. Delhi High Court granted interim injunction, emphasising danger to diabetic patients.[16]
8.5 Glenmark Pharmaceuticals v. Curetech Skincare
CANDID-B versus CLODID-B: Both marks for antifungal/dermatological preparations (Class 5). Phonetically similar in clinical or pharmacy settings; similar packaging colours contributed to likelihood of confusion. Bombay High Court granted injunction in favour of Glenmark.[17]
8.6 Abbott Healthcare v. Vinsac Pharma
LIMCEE® versus LIMEECEE: Abbott’s established Vitamin C supplement brand LIMCEE®. The infringing mark LIMEECEE incorporated LIMCEE entirely, being structurally and phonetically identical. Delhi High Court granted injunction.[18]
8.7 Mankind Pharma v. Sepkind Pharma
MANKIND® versus SEPKIND: Mankind Pharma’s well-known mark versus a defendant that incorporated the ‘-KIND’ suffix, deliberately riding on Mankind’s goodwill. Visual logos showed similar colour scheme, font style, and arrangement. Delhi High Court granted injunction, noting mala fide intent in the choice of corporate name.[19]
8.8 Mankind Pharma v. Sanshiv Health Tech
CALIKA-P versus CALDIKIND-P: Both for Calcium + Vitamin D3 + Vitamin K combination supplements, identical therapeutic composition. Both marks began with ‘CALI/CALDI’ and ended in ‘-P’. Court found phonetic and structural similarity; injunction granted.[20]
8.9 Johnson & Johnson v. Pritamdas Arora
A counterfeiting case involving fake J&J-branded bandages and medical devices sold through medical supply distributors. The Delhi High Court in 2025 granted a permanent injunction, awarded enhanced damages, and ordered delivery-up and destruction of infringing goods.
9. Trademark Enforcement: Civil and Criminal Remedies in India
The Trade Marks Act, 1999 provides a comprehensive enforcement framework. In pharma, where health consequences are immediate, rights holders typically seek urgent interim relief before proceeding to full trial.
Civil Remedies
- Temporary Injunction: Immediate interim relief to stop infringing sales pending trial – the most critical first step in any pharma enforcement action.
- Permanent Injunction: Final restraining order after full trial.
- Anton Piller Order: Ex-parte court-authorised search and seizure of infringing stock – available in urgent cases.
- Damages / Account of Profits: Plaintiff may elect either actual loss or the profits made by the infringer.
- Delivery Up and Destruction: Court orders infringing goods and labels to be delivered up and physically destroyed.
Criminal Remedies
- Section 103, Trade Marks Act, 1999: Imprisonment between 6 months and 3 years; fine between Rs. 50,000 and Rs. 2,00,000.[22]
- Police Powers: Police may act without warrant with the Trademark Registrar’s approval.
- Drugs & Cosmetics Act, 1940: Additional criminal liability for spurious and misbranded drugs; imprisonment up to life imprisonment in cases where the patient dies.
- Border Measures: Customs seizure under the IP Rights (Imported Goods) Enforcement Rules, 2007.
- Online Enforcement: Court orders directing platforms such as Amazon and Flipkart to delist counterfeits; John Doe / Ashok Kumar orders for unknown defendants.
Conclusion: Trademark Strategy as Competitive Advantage in Pharma
In the pharmaceutical and biotechnology industries, trademark strategy is not a post-patent afterthought – it is the most durable commercial weapon a company can deploy. The data and case law reviewed in this article converge on five conclusions:
- File First, Launch Later. Covaxin® and Ozempic® in India demonstrate the critical importance of filing trademarks – including via Madrid Protocol – before entering new markets or announcing products.
- Brand Outlives Patent. Humira®, Crocin®, and J&J’s brand portfolio demonstrate that a strong trademark sustains market position and revenue long after the patent expires.
- Clearance Is Not Optional. The consequences of a post-launch rebrand in pharma are catastrophic: wasted packaging costs, lost marketing investment, CDSCO re-approval delays, and potential injunctions.
- Courts Apply Strict Standards. Indian courts have consistently held that even partial phonetic similarity between pharmaceutical marks is sufficient to establish deceptive similarity and grant injunctions.
- Platform Branding Is the Future. Keytruda® and Moderna’s mRNA platform demonstrate that biotech companies now brand technologies and platforms, not just products.
Our Coverage on LinkedIn: https://www.linkedin.com/feed/update/urn:li:activity:7457297240521363456
[1] “Pharmaceuticals Industry in India,” IBEF Sector Report, 2024-25: https://www.ibef.org/industry/pharmaceutical-india
[2] https://www.ibef.org/industry/pharmaceutical-india.
[3] Pharmaceutical Market Size, Share and Trends 2024 to 2034”
[4] “Substandard and Falsified Medical Products,” WHO Fact Sheet (January 2018, updated 2023): https://www.who.int/news-room/fact-sheets/detail/substandard-and-falsified-medical-products
[5] “Illicit Trade: Converging Criminal Networks” (2016): https://www.oecd.org/corruption/illicit-trade-9789264251847-en.htm.
[6] Drug Alert Notices 2024: https://cdsco.gov.in/opencms/opencms/en/Alerts/Drug-Alerts/
[7] Trade Marks Act, 1999, Section 2(1)(zb)
[8] Trade Marks Act, 1999, Section 13: INN Programme: https://www.who.int/teams/health-product-and-policy-standards/inn.
[9] World Health Organization, INN Programme: https://www.who.int/teams/health-product-and-policy-standards/inn.
[10] WHO INN Programme Guidance Document, 2013; WHA Resolution 46.19 (1993); Trade Marks Act, 1999 – Section 13.
[11] US Food and Drug Administration, “Nonproprietary Naming of Biological Products: Guidance for Industry” (January 2017)
[12] FDA Guidance for Industry – Nonproprietary Naming of Biological Products (2017); WHO INN Programme; FDA Purple Book.
[13] Cadila Healthcare Ltd. v. Cadila Pharmaceuticals Ltd., (2001) 5 SCC 73 (Supreme Court of India)
[14] M/s Abbott GmbH v. Registrar of Trademarks, C.A.(COMM.IPD-TM) 9/2023 (Delhi High Court, 2023)
[15] Sun Pharmaceutical Industries Ltd. v. Anglo French Drugs and Industries Ltd., FAO(OS) 66/2014
[16] Glenmark Pharmaceuticals Ltd. v. Gleck Pharma (OPC) Pvt. Ltd., 2024 SCC OnLine Del 1660
[17] Glenmark Pharmaceuticals Ltd. v. Curetech Skincare and Others, 2018 (76) PTC 114 (Bombay High Court, 2018)
[18] Abbott Healthcare Pvt. Ltd. v. Vinsac Pharma and Others, MANU/DE/1313/2025 (Delhi High Court, 2025).
[19] Mankind Pharma Ltd. v. Sepkind Pharma Pvt. Ltd. and Others, 2024 SCC OnLine Del 8982 (Delhi High Court, 2024)
[20] Mankind Pharma Ltd. v. Sanshiv Health Tech Pvt. Ltd., CS(COMM) 418/2024 (Delhi High Court, 2024).
[21] Johnson and Johnson v. Pritamdas Arora and Others, 2025 SCC OnLine Del 1504 (Delhi High Court, 2025).
[22] Trade Marks Act, 1999, Section 103


