PATENTS
Compulsory licence -no longer an unmentionable word
Natco continue to remain India’s first Compulsory License Holder as IPAB upheld Controller’s Decision (March 4, 2013) The Indian Intellectual Property Appellate Board (IPAB) rejected appeal filed by Bayer Corporation (Appellant) against the Controller General’s decision granting Compulsory Licence to Natco Pharma Ltd. (Respondent) for Bayer’s patented drug Nexavar (Sorafenib Tosylate). Earlier in 2012 the Indian Patent Office granted its first Compulsory License to Natco for producing generic version of Bayer’s patented medicine Nexavar (Sorafenib Tosylate), which is used in the treatment of Liver and Kidney cancer. While the multinational giant was selling the drug at INR 2.80 lakh for a month’s course, Natco promised to make available the same at a price of about 3 % (INR 8800) of what was charged by Bayer. Aggrieved by the Controller’s decision, Bayer had immediately moved to the Intellectual Property Appellate Board for stay on the order alleging that the grant of Compulsory License was illegal and unsustainable. The Board rejected Bayer’s appeal holding that if stay was granted, it would definitely jeopardize the interest of the public who need the drug at the later stage of the disease and that granting stay would affect the right of access to affordable medicine of the patients.
The IPAB rejected all the contentions raised by Bayer in their appeal including notice not being given to appellant (opportunity at the prima facie stage), genuine attempts not being made by the Respondent to secure licence from Appellant, reasonably affordable price and working of patented invention in the territory of India.
Interestingly Bayer was banking on Cipla’s presence in the market and had contended that CIPLA has been selling the disputed drug at Rs 30,000 and until declared as an infringer, its presence cannot be considered illegal and should be taken into reckoning to see if the reasonable requirement of the public is met.
The Board held that the Controller was right in holding that the sales of the drug by the appellant at the price of about 2,80,000/- was alone relevant for the determination of public requirement and he was also right in considering the purchasing capacity of the public and the evidence available to conclude that the invention was not reasonably affordable to the public.
It held that with regard to Section 84(1)(c), it finds that the word ‘worked’ must be decided on a case to case basis and it may be proved in a given case, that ‘working’ can be done only by way of import, but that cannot apply to all other cases. The patentee must show why it could not be locally manufactured. A mere statement to that effect is not sufficient there must be evidence.
On the percentage of royalty that was to be paid by the Respondent to the Appellant (6% that was fixed by the Controller), IPAB increased it by 1 percent but did not change any other terms and conditions of the licence.
The IPAB dismissed the appeal and confirmed the grant of Compulsory license stating that it had dealt with each of the issue in detail in view of the significance of the order of compulsory licence made in India for the first time.
Section 3-d continues to rule as India’s Premier Court Rejects Novartis’s Appeal (April 1, 2013).
In what would be touted as the landmark judgment in all the years to come for the Pharma Industry as well as the Indian Patenting System, the Supreme Court of India rejected Novartis’s plea on its blood cancer drug Glivec and denied patent to it. The drug was not considered to be patentable as per the provisions of Section 3(d) that draws a distinction between “ever-greening” and incremental innovation.
The Supreme Court rejected the claims of Swiss pharma giant stating that the drug had failed to qualify for a patent according to Indian law. The Court also held that patents could be granted only in the case of genuine inventions. It is worth mentioning that Gleevec is approved in more than 90 countries including the US, EU, and Japan for the treatment of all phases of Ph+ CML.
What is non-patentable under Section 3(d):
(d) the mere discovery of a new form of a known substance which does not result in the enhancement of the known efficacy of that substance or the mere discovery of any new property or new use for a known substance or the mere use of a known process, machine or apparatus unless such known process results in a new product or employs at least one new reactant.
The underlying assumption of section 3 (d) is that derivative, such as salt forms, polymorphs, isomers etc that are structurally similar to known pharmaceutical substances are likely to be functional equivalent as well, and if this is not the case and the new form of an existing substance works better than the old form, it is up to the patent applicant to demonstrate this and justify the claim to a patent.
Novartis had been fighting since 2006 to get the patent for Glivec, which as claimed by oncologists is major advance in treating chronic myeloid leukaemia. It had been continuously pushing for the patent arguing that if the rights of investors are not upheld then it will hit research and development of new drugs. The Supreme Court held that the prior patents and literature did count as prior art against the beta crystalline form of imatinib mesylate and it did not meet the requirements of an “invention” as laid down in the Indian Patents Act.

Regarding efficacy the Court held that “Efficacy means the ability to produce a desired or intended result. Hence, the test of efficacy in the context of section 3(d) would be different, depending upon the result the product under consideration is desired or intended to produce. In other words, the test of efficacy would depend upon the function, utility or the purpose of the product under consideration. Therefore, in the case of a medicine that claims to cure a disease, the test of efficacy can only be ‘therapeutic efficacy’.” The Court also held that the flow properties, thermodynamic stability and hygroscopicity, on which Novartis was relying to overcome the objection under s 3 (d), had nothing to do with therapeutic efficacy. On Novartis’s claim on increased bioavailability, the Court held that increase in bioavailability of the beta crystalline form was not in comparison with the known and previously marketed salt form of the drug which was soluble, but rather in comparison to the free base form which was not marketed as it was not highly soluble. The Court therefore rejected the Novartis patent application as increased bioavailability was not proven and efficacy test of s. 3(d) was not met. The Supreme Court had also held that the novelty in the said case was not proven on facts and that the drug would have failed the patentability test on this basis alone.
GSK cancer drug patent on Tykerb revoked by IPAB (July 27, 2013)
FRESENIUS KABI ONCOLOGY LIMITED initiated the rectification proceedings against GLAXO’s two patented drugs Bicyclic Heteroaromatic Compound (Patent No. 221017) and Quinazoline Ditosylate Salt Compounds [Patent No. 221171]. The patents were for Glaxo’s anti-cancer (breast cancer) drug Tykerb. Similar grounds were raised in revocation proceedings of both the patents, i.e. of obviousness, new form of a known substance (Section 3 (d)) and non disclosure of corresponding foreign applications (as that required under Section 8 of the Indian Patent Act.
The IPAB observed that in the Novartis case, the Supreme Court had held that physico-chemical properties had nothing to do with therapeutic efficacy. Applying Section 3(d) of the Patent Act and the Supreme Court’s decision, IPAB revoked the latter patent. The revocation proceedings for the former patent were however rejected by the Intellectual Property Appellate Board.
The interesting factor being deliberated by IPAB in both the proceedings was the requirements of Section 8. Though FRESENIUS’s objections regarding non compliance of Section 8 was rejected in both cases due to their failure to plead the facts and state how the particular undisclosed application was for the same or substantially the same invention, the Board took a tough stand on it. So much so that the Board noted: To the question “Why should we comply with S.8?” The Answer is “Because it is there.”
Merck Sharp and Dohme denied Injunction by Single Bench, Division Bench reserved verdict (August 1, 2013, April 5, 2013)
In yet another dispute surrounding pharma patents in India, US pharmaceutical giant Merck Sharp and Dohme (MSD) battled against Mumbai based local drug maker Glenmark Pharmaceuticals Limited over its anti-diabetic patent drug comprising the molecule SITAGLIPTIN. The interesting fact about Merck’s case is that, pricing and hence the availability of the drug was not the issue in the present case, since in India the drug is roughly priced at 1/5th of its price in the USA, the battle for injunction was purely on technological grounds. Merck itself claimed that this was probably the first case of differential pricing in India and that the price was arrived at after surveys, and that the same is within the reach of the people in need of the said medicine. Merck had alleged that the Indian pharma company had violated its IPR over its anti-diabetes medicines, Januvia and Janumet, by coming out with their own drugs containing the same salts.
Merck contended that Glenmark being a large pharmaceutical company was well aware of its product JANUVIA and its active ingredient as well as of the patent granted to cover the same. Merck also stated that Glenmark itself obtained US patent (dated 18th December, 2012) for its process for preparation of SITAGLIPTIN and, in the said patent, has acknowledged Merck’s corresponding US patent for SITAGLIPTIN and its proprietary rights by making admissions thereof.
Merck further remarked that Glenmark had started distributing SITAGLIPTIN and SITAGLIPTIN PLUS METFORMIN (under the brand names ZITA and ZITA-MET) in the form of sample packs prior to the institution of the suit and has hurriedly dumped the product in the market to gain mileage in any law-suit instituted against them; and had filed caveats with the same intent.
Glenmark on the other hand stated that Merck had not disclosed that it had applied for grant of patent of the product qua which injunction is claimed, which was not only declined but was also affirmatively abandoned by Merck; Glenmark contented that Merck was guilty of suppression and should not be entitled to the grant of discretionary relief of interim injunction on this ground alone.
The Single Bench while denying the infringement noted that in the present case, the plaintiff Merck as patentee of SITAGLIPTIN is not marketing SITAGLIPTIN alone as a product and is marketing Sitagliptin in combination with Phosphate just as the defendant is doing. However as the defendant pointed out that the patent of the plaintiff Merck was not in the pharmaceutical composition (as described on plaintiff’s product) but only in a part thereof and which fact was not denied by the plaintiff. Thus, similarity of pharmaceutical composition of the products cannot be a ground for infringement.
Thereafter an appeal was filed by the US drugmaker in the Delhi High Court. A bench of justices S.Ravindra Bhat and Najmi Waziri however reserved the judgement after the counsel for both the drug manufacturing firms concluded their arguments in the case.
Allergan Inc.’s patents on eye drugs Ganfort and Combigan revoked by IPAB (August 8, 2013)
The Intellectual Property Appellate Board revoked two of Allergan, Inc.’s patents IN 212695 and IN 219504 which were granted by the Kolkata Patent Office in December 2007 and May 2008 respectively. Both the patents were related to drugs used in the treatment of Ocular Hypertension (Glaucoma). The rectification applications were filed by Ajanta Pharma Limited a specialty pharmaceutical company headquartered in Mumbai and engaged in development, manufacture and marketing of quality finished dosages in domestic and international markets. The grounds of rectification application were obviousness, not an invention, not patentable, insufficiency and non-compliance of Section 8 of the Indian Patents (Amendment) Act, 2005.
Disputed Patent no. 212695 covered the drugs Ganfort and Combigan which are used in the treatment of ocular hypertension. The said patent is a fixed combination of Bimatoprost and Timolol. Allergan claimed to achieve enhanced treatment of ocular hypertension with reduced side effects. At the very outset Allergan challenged the revocation of the impugned patent by Ajanta stating that the revocation proceedings were time barred. It also submitted that the claimed invention provides compositions which are surprisingly effective in treating ocular hypertension and reducing die number and/or frequency and/or severity of unwanted side effects caused by die bimatoprost component, relative to prior art compositions and methods.
The Board observed that in the instant case Allergan had failed to comply with the requirements as entailed under Section 8 as no information relating to prosecution of same application in other countries was provided. It also held the invention to be obvious stating that Latanoprost is not different from Bimatoprost. It further stated that Allergan may have been easily led to believe by prior art teachings that Bimatoprost like Latanoprost is capable of producing the same result if not better. In view thereof, the choice of Bimatoprost to replace Latanoprost was obvious.
Further the Board rejected Allergan’s contentions regarding revocation application being time barred stating that it was not barred by Limitation as the right to revoke patent any time after the grant of patent under section 64 cannot be extinguished after three years from the date of the publication of the grant by applying limitation of three years under Article 137 of the Limitation Act as the same would run contrary to the scheme of the patent law.
Disputed Patent no. 219504 held by Allergan was claimed to be a combination of two active drugs namely Brimonidine and Timolol proposed for administration in ophthalmic conditions. Allergan contended that U.S. FDA approved Brimonidine to be administered thrice a day and Timolo twice a day. The serial administration of the two drugs was also known but the combination of two drugs in a fixed combination was never taught or suggested by a prior art. Thus, the combination is an inventive step. It demonstrated that the fixed combination, alternative dosing regimen would provide a useful product because the safety profile of the proposed combination is better than that of the individual agents taken as currently permitted in the approved labelling.
The Board observed that Allergan inspite of possessing the final office actions as issued by the EPO and USPTO did not submit them thus violated Section 8 of the Act. With respect to Section 3 (d) the Board rejected Ajantha’s plea and held Allergan right on the ground that the combination of Brimonidine and Timolol cannot be considered as derivatives of each other. Regarding obviousness of the invention, the Board was of the view that in the history of the State of the Art indicated that the two drugs were popular and that they were combined serially and that the serial administration of the two drugs showed advantages over single therapy. The Board in this regard further observed that the drug Brimonidine was not unknown and that the composition of the two drugs in one bottle was also known and the claimed preservative was also used thus indicating the invention as an obvious one.
In view thereof the Board revoked both the impugned patent.
IPAB revoked Kibow Biotech’s Patent (November 13, 2013)
Kibow Biotech Inc., a fourteen year old biotechnology company specializing in the development and commercialization of scientifically formulated and clinically tested probiotic dietary supplements lost one of its patents on process of Making Pharmaceutical Composition. The IPAB revoked the said patent as it was found not to comply with the requirements of the Indian Patent Act, especially Section 3(e).
The Gujarat based La Renon Health Care Pvt Ltd. was marketing its product under the brand name CUDO. Kibow issued a cease and desist notice upon them (on October 5, 2010) calling upon them to restrain from infringing its patent in their product “CUDO”. In its reply statement La Renon contended that it believes that the composition and process of manufacturing of their product CUDO is unique, superior and first of its kind in India which cannot be disclosed as it is in the process of getting the patent. Kibow thereafter requested for the claimed patent application and the date of filing of the said application. As no information about its patent was furnished and La Renon did not restrain itself from manufacturing and selling the product CUDO, a Civil Suit C.S. No.498/2011 before the Hon’ble Madras High Court was filed by Kibow Biotech alleging infringement of patent Nos. 205478 and 224100. The said suit was dismissed by the High Court (vide order dated August 22, 2011) due to non-maintainability of the suit under Section 599 of the Companies Act as per the provisions of Order 7 Rule 13 Code of Civil Procedure. The Hon’ble Court however said the plaintiff (i.e. Kibow Biotech) was entitled to bring a fresh suit after curing the defects of Section 599 of the Companies Act.
Thereafter the revocation application, against the grant of patent nos. 224100 and 205478 was filed before the IPAB by La Renon Health Care Pvt Ltd. While application for patent no. 224100 was dismissed by IPAB, patent no. 205478 was revoked for failing to meet the patentability criteria- novelty and obviousness and falling within ambit of Section 3(e).
With respect to Patent No. 224100 the IPAB considered all the granted claims and on comparing the same with the cited documents held that none of the limitations of the granted claims were taught in any of the cited document either alone or in combination. It held that mere speculation of a hypothesis is not enough to defeat the claimed invention on the ground of obviousness. On compliance with Section 3 (e) IPAB accepted Kibow’s argument and held that the composition comprises at least one probiotics bacteria of a certain kind, in a certain composition and it is not a mere admixture.
However with respect to Patent No. 205478, IPAB noted that as proportions of mixing of various components of the invention were not apparent from the examples, the synergistic effect was not outcoming. It held that held that the invention is obvious based on the prior art as the use of each of the integers, as claimed in the invention, and micro encapsulation or enteric coating are already known from the background of the invention. There is no interworking between the integers to result in a synergistic effect.
IPAB evoked the patent holding that the claimed pharmaceutical composition containing five integers is neither a new product, nor the mixing of the products advance any technical effect over the existing prior art. Thus, it makes the invention obvious to a person skilled in the art. It was also held that the mixing of the five integers is only a mere admixture and that there was no invention as per the provisions of the Act.
BDR Pharmaceuticals’ application for Compulsory License for Bristol-Myers cancer drug rejected by the Indian Patent Office October 29, 2013)
The Controller General of Patents in India rejected Mumbai based BDR Pharmaceuticals International Pvt. Ltd’s application for compulsory license for Bristol-Myers cancer drug SPRYCEL (active pharmaceutical ingredient DASATINIB – Patent number 203937) stating that BDR Pharmaceutical failed to make out a prima facie case for the making of an order under section 87 of the Act.
BDR Pharmaceuticals had requested for compulsory license earlier this year (on March 4, 2013) claiming that DASATINIB is a suitable chemotherapeutic option for the treatment of CML and is prescribed when a patient is resistant or develops resistance to the drug IMATINIB, in view of the improved tolerance and efficacy of the drug.
It was further claimed that DASATINIB is administered as 50 mg tablets with a dosage of 100 mg per day and thus, two tablets are to be consumed per day until disease progression or until the patient can no longer tolerate the medicine. Based on this premise BDR Pharmaceutical stated that the price of each tablet sold by the patentee is INR 2761 /- which works out to INR l, 65,680/- for 60 tablets per month per patient and about INR 19,88, 160/- per year per patient.
BDR claimed in its application that it would make the drug available at INR 8100 per month for the treatment of a CML patient and that it would be offering the drug free of cost to a certain percentage of patients suffering from CML as determined by the cancer specialists.
In the instant case BDR had sent a letter dated February 2, 2012, to Bristol Mayer requesting for a voluntary license for manufacturing DASATINIB. By letter dated 13th March 2012, the patentee raised certain queries such as “facts which demonstrate an ability to consistently supply high volume of the API, DASATINIB, to the market “, “facts showing BDR’s litigation history or any other factors which may jeopardize Bristol-Myers Squibb’s market position “, “quality related facts and in particular compliance with local regulatory standards and basic GMP requirements”, “quality assurance systems due diligence”, “commercial supply teams”, “safety and environmental profile “, “risk of local corruption”.
BDR took this reply of the patentee as “clearly indicative of the rejection of the application for voluntary license and did not pursue the matter.” Thereafter on March 4, 2013 BDR filed an application with the Patent Office for grant of compulsory license.
The Controller in his decision, while rejecting the application, stated that the decision to grant a voluntary license, particularly on a subject matter covered by a patent, is an important decision for a patentee. While, it is possible that some of the queries raised by the patentee may not be strictly reasonable, it is natural that the patentee may seek additional information from the requesting party to satisfy himself about the credentials and capability of the said party.
It is pertinent to mention that on BDR’s assumption of queries raised by the Patentee as indicative of rejection of the application for voluntary licensee theController stated that “On the face of the record, I am of the view that the applicant’s contention that the said letter is ‘clearly indicative of the rejection of the application for voluntary license does not hold good, as the aforementioned queries raised by the patentee appear largely to be reasonable” and that the Applicant ought to have appreciated that the provisions relating to compulsory license are to be invoked as the last resort, i.e. if the mutual deliberations do not lead to a result within six months, in accordance with the scheme of the law.
The Controller held the applicant did not make efforts to obtain a license from the patentee on reasonable terms and conditions and therefore rejected the said application.
The TRADEMARKS
The Long- Awaited Accession to Madrid Protocol
One of the most significant achievements of India in the year 2013 with respect to Intellectual Property Rights was the long anticipated accession to Madrid Protocol which finally materialized on April 08, 2013 when Mr. Anand Sharma, Minister of Commerce and Industry, handed over India’s instrument of accession to WIPO Director General Francis Gurry. The Treaty came into effect in India on July 08, 2013.
The Madrid System would enable trademark owners in the signatory countries to secure International Registration by way of filing a single application. One of the most significant advantages of International Registration of Marks is that a person applying for a mark through the Madrid System has to file only one application, in one language, with one set of fees (including basic fee, complimentary fee in respect of ach designated contracting party and a supplementary fee for each class of goods beyond the third) in one currency. Moreover, the expiry date with respect to all the designated countries will be one and the mark can be renewed only through a single application.
Legislative and Administrative Reforms Initiated in India.
India made legislative reforms and aligned its Intellectual Property regime with the global norms through the Trademark (Amendment) Bill, 2009 which led to the inclusion of a new chapter (Chapter IV A) in the Trademarks Act. The said Chapter exclusively deals with procedural and statutory requirements for international registration of marks. Sections 36A to 36G of the Trademarks Act entails the provisions with respect to international registration of marks under the Madrid System. Under this provision on receipt of notification from International Bureau with respect to a particular mark, the Registry will examine the mark under the provisions laid down under Sections 9, 21, 63 and 74 of the Act and thereafter accordingly issue necessary directives (advertise or refuse) the application and apprise the International Bureau with respect to the same within 18 months from the date on which it received the notification.
It would be pertinent to mention here that pursuant to accession to Madrid System, the Indian Trademarks Registry has endeavoured to raise awareness regarding the same and has recently been conducting several conferences and seminars all over the Indian territory with the object to raise awareness among applicants and agents regarding the system, process and implementation of the International Registration of Marks. The Trademarks Registry has also recently formulated and published the Guidelines for functioning under the Madrid Protocol
and the same can be accessed at http://ipindia.nic.in/Whats_New/guidelines_MadridProtocol_17December2013.pdf .
Expected Impact of International Registration of Marks on the Domestic Market
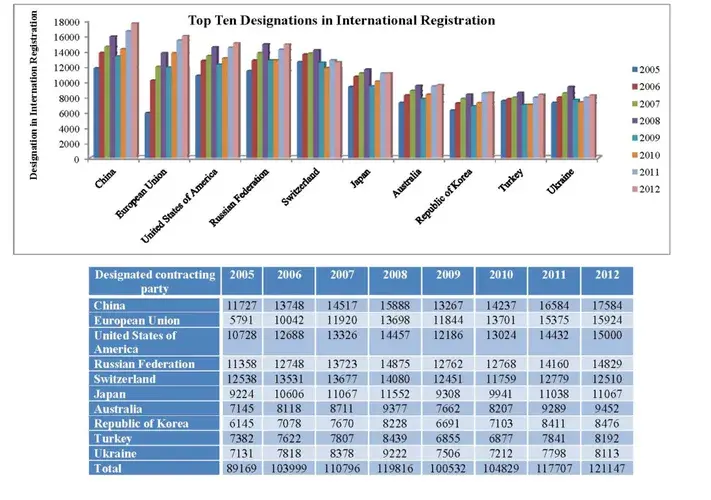
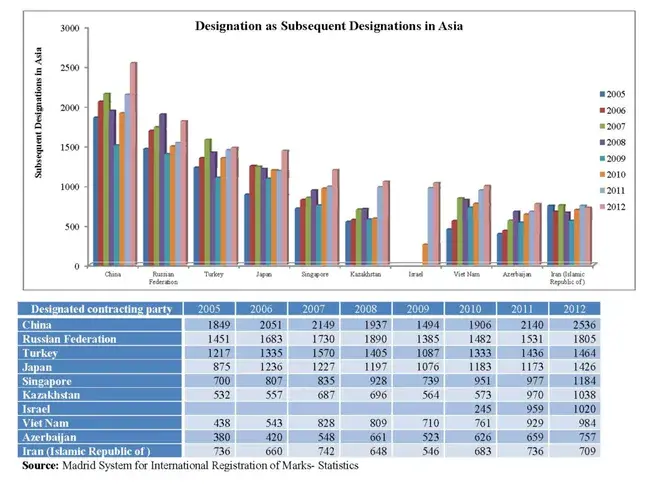
It is being widely speculated that the Madrid System of International Registration of Marks would lead to a gradual decline in Indian Trademark Applications; however the statistics (as made available by Madrid System for International Registration of Marks) reflect that the filing and registration of marks through the Madrid System has rather augmented in the past few years. Moreover, India being figured as one of the most desired consumer markets in the recent times is likely to be more often designated by foreign applicants via the Madrid Protocol.
Graphs demonstrating acclivity in filing through the Madrid System worldwide and in Asia is depicted below:
The Obfuscating Saga of Comparative Advertising in India
Year 2013 can be remarked as one of the years’ which saw several cases pertaining to comparative advertising being filed and law being sometimes defined, re-defined, sometimes dissented and at times being overruled by the Indian Judiciary which subsequently left business organizations and common public perplexed regarding the ethical and non- ethical aspects of comparative advertising in India.
Reckitt Benckiser and Hindustan Lever could be perceived as two companies which were on a spree of airing comparative advertisements and subsequently approaching the Court alleging product disparagement against each other.
Advertisements by Reckitt Benkiser (India) Ltd.:
Advertisement I:
This advertisement through print media showed ‘Dettol Healthy Kitchen Gel’ was capable of killing 100X more germs as compared to any ‘leading dish-wash’. The said advertisement however did not show “VIM” anywhere.

Taking into consideration the facts and circumstances of the aforesaid advertisement, the Court was of the view that though the said advertisement does not show the competing product as ‘VIM’, the reference to a ‘leading dish-wash’ would lead any reasonable person to think that the dish-wash being referred to was ‘VIM’ as VIM enjoys about 2/3rd share of the market and the other dish-washes available in the market are quite insignificant as compared to ‘VIM’.
The Court further opined that proclaiming that ‘Dettol Healthy Kitchen Gel’ kills 100X more germs is permitted but showing that application of Dettol Healthy Kitchen Gel nearly killed 100% of the germs whereas application of ‘VIM’ killed no germs, certainly denigrated ‘VIM’ and lowered its reputation. Therefore, the said advertisement was hit by Section 30 (as reproduced below) of the Trade Marks Act, 1999.
Limits on effect of registered trade mark
- Nothing in section 29 shall be construed as preventing the use of a registered trade mark by any person for the purposes of identifying goods or services as those of the proprietor provided the use –
- is in accordance with honest practices in industrial or commercial matters, and
Advertisement II:
In this advertisement Reckitt has clearly compared its product Dettol Healthy Kitchen Gel to Hindustan Lever’s product by showing the mark of its competitor i.e. VIM specifically mentioned in the commercial.

With respect to the aforesaid advertisement, the Court was of the view that the same reflected a direct comparison of the two products i.e. Dettol Healthy Kitchen Gel and VIM. The impugned advertisement showing that Dettol Healthy Kitchen Gel kills 100X more germs than the ‘leading dish-wash’ or VIM indicated that the content of the advertisement suggested a serious comparative advertisement. The Court was of the view that though comparative advertisements are safeguarded under the law of commercial speech which is protected by Article 19 (1) (a) of the Constitution there are still restrictions on such commercial speech which are “deceptive, unfair, misleading and untruthful.”
Advertisements by Hindustan Unilever (India) Ltd.:
Advertisement III:
In the said advertisement Hindustan Lever showed that Lifebuoy soap gave better germ protection than Reckitt’s Dettol antiseptic liquid.

On hearing the contentions of both the parties and analysing the impugned advertisement the Court was of the view that the advertisement made an unfair comparison between two products which were apparently not meant for the same purpose. Dettol is an antiseptic liquid that is predominantly used for germ eradication and is used as a supplement to soap/ bathing gel while Lifebuoy is used predominantly for cleansing.
The Court observed that showing the plaintiff’s product in an inappropriate perspective and then, declaring that it has no germ protection capacity whereas the defendant’s product ‘Lifebuoy Soap’ had nearly 100% germ removal capacity, was disparaging the defendant’s product.
Advertisement IV:
This advertisement showed that an antiseptic liquid is unsafe for children and hence utensils should not be washed with them. Moreover, VIM was projected as a safe cleaner, having natural ingredients like lemon to clean utensils.

In reference to the aforesaid advertisement, the Court was of the view that the same did not directly refer to Reckitt’s product Dettol as it showed a bottle with an amber coloured liquid termed as an antiseptic. However, the picture of the bottle with an amber liquid, even in the little plot of the advertisement, would convey to any reasonable person, having knowledge of Dettol Antiseptic Liquid and dishwash that the said bottle referred to Dettol Antiseptic Liquid.
The Court further observed that the advertisement’s reference to the dangerous nature of antiseptics on consumption had a real possibility of deterring consumers from purchasing the Dettol Kitchen Gel. Thus, the said advertisement was also categorized as a comparative advertisement implicating serious comparison of two products of two manufacturers and declaring one to be safe and superior.
Another case which made to the Court with issue relating to comparative advertising was Marico Limited vs Adani Wilmar Ltd. (CS(OS) 246/2013) wherein Marico was marketing its cooking oil under the brand name Saffola, and approached the Court with the request for a permanent injunction to restrain Adani Wilmar from broadcasting, publishing and printing advertisement of its product under the brand name Fortune.

The High Court of Delhi, while arriving at its decision, took into consideration a plethora of cases where Courts have at length dealt with the issue of comparative advertising. The basic submissions of Marico v Adani Wilmar were that its product, Saffola, had allegedly been disparaged by television commercials and print advertisements issued by the defendant (Adani Wilmar), by making blatantly false, unsubstantiated and misleading claims with respect to its cooking oil sold under the brand name Fortune RBO (rice bran oil) as being the healthiest oil in the world, healthier than the plaintiff’s cooking oil Saffola. The plaintiff also contended that Adani Wilmar had deliberately displayed the disclaimer in its advertisement in such a manner that it was not visible to the audience, and also appeared to be vague and misleading.
The Court, in arriving at its decision, made reference to the cases of De Beers Abrasive v International General Electric, and the decision of the Division Bench (Delhi High Court) in the case of Dabur India v Colortek Meghalaya, and held that Adani Wilmar’s impugned advertisement was not disparaging. It observed that Adani Wilmar only compared the advantages of its own cooking oil over the advantages of cooking oil marketed by others.
Another case which has apparently wound up the issue relating to comparative advertisements for the year 2013 is the case of Colgate Palmolive (India) Ltd. v. Hindustan Unilever Ltd. (CS (OS) No. 1588 of 2013).

In the said case the Delhi High Court on August 21, 2013 had delivered its order in favour of comparative advertising. In the said case the grievance of the Plaintiff was that the impugned advertisement misused the Plaintiff’s registered trademark Colgate and also disparaged the goodwill and reputation of its toothpaste namely Colgate Dental Cream Strong Teeth and the said advertisement tarnished the reputation of Plaintiff’s brand. Colgate contended that the claim made by the Defendant that Pepsodent Germicheck had 130% attack power was false which apart from violating the ASCI code also violated the Drugs and Cosmetics Act. The Plaintiff further contended that its product in the ad was shown in a bad light as it showed that the use of Colgate could cause cavities.
The Court while arriving at its decision analysed the principles of Comparative Advertising as laid down in the case of Dabur India Ltd. vs M/S Colortek Meghalaya Pvt. Ltd. and was of the view that the impugned ads were not disparaging and too much could not be read into the ads and to the expressions of each individual character. The Court further observed that the expressions and effects used in the ad, only showed that Pepsodent was a better product and did not disparage Colgate’s product.
Aggrieved by the aforesaid judgement delivered by Single Judge of the Delhi High Court Colgate filed an appeal. The case was taken up by the Division Bench of the High Court on December 10, 2013 which ruled that the impugned advertisement could not per se be considered as disparaging. The Division Bench further pronounced that the voice over at the end of the impugned television campaign which stated that NAYA PEPSODENT GERMI CHECK COLGATE KE MUKABLE 130 PRATISHAT GERM ATTACK POWER was misleading and inaccurate. Pepsodent was directed to delete the same from the impugned television campaign or that at the option of Colgate, may be suitably modified to refer only to COLGATE STRONG TEETH and not COLGATE.
The Division Bench further restrained Pepsodent from publishing the impugned print advertisement or any other similar advertisement which would disparage COLGATE STRONG TEETH or any other product of Colgate.
In view of comparative advertisement cases taken up by the Judiciary and judgments pronounced therein, it can be inferred that the advertisers have to be very careful while creating an ad and analyze various ethical and legal aspects of comparative advertising wherein while comparing one’s product with competitor’s product it can show that its product is better but cannot denigrate the competitor’s product.
Adieu to Trademarks of The Well Known Brands- Nirma, Cadbury, Kit Kat, Maaza
In the case of M/s Nirmal Industrial Controls Pvt. Ltd.v. M/s Nirma Chemical Works Pvt. Ltd., the Intellectual Property Appellate Board (IPAB) ordered removal of the famous trademark NIRMA from the Register. Downloader java script – code image capture

In the said case, the Plaintiff i.e. Nirmal Industries had filed trademark application for the mark ‘NIRMAL with device” “process control valves, instruments systems” falling under class 6 and claiming use since April 01, 1973. On being advertised the impugned mark was opposed by Nirma on the grounds that the mark NIRMAL was deceptively similar to its registered mark NIRMA in class 6. The Registry on the basis of opposition Refused Plaintiff’s mark. Aggrieved by the order of the Registry, the Plaintiff approached the IPAB.
The IPAB on hearing the contentions of both the parties and examining the evidence adduced in the case was of the view that NIRMA was unable to establish use of its mark in class 6 and that none of the documents produced by them reflected that there was a bonafide intention to use the mark NIRMA in class 6. While removing NIRMA’s mark on the grounds of non- use the IPAB also questioned the conduct of the Registry in the instant case and remarked that an order shall rely on the merits of the case and not be a mere emissary of well- known marks.Removal of Cadbury’s Three Marks- In response to the rectification proceeding initiated by ITC Ltd., the IPAB recently passed an order for removal of three of Cadbury’s trademarks namely- Cadbury’s Chocolate Eclairs (a label mark), CADBURY’S CHOCOLATE ECLAIRS (a label mark) and Cadbury’s Orange Flavoured Chocolate Eclairs (a label mark) on the grounds of non- use which were registered in class 3.

The tussle between ITC and Cadbury commenced when ITC received an ex parte injunction order on April 01, 2005, restraining it to use the mark ECLAIRS or any other deceptively similar marks. Aggrieved by the said order ITC approached the Gujrat High Court wherein the Court modified the earlier order and held that ITC can manufacture and sell their Eclairs products as Candyman Choco Eclairs. Thereafter, ITC approached the IPAB for rectification of Cadbury’s marks.
Cadbury contended that it introduced the product under the name Cadbury Chocolate Eclairs/Eclair in the year 1972 in India and registered the trade mark Cadbury Chocolate Eclairs (vide registration no.298102 in Class 30) as early as on August 1, 1974, in respect of Milk Chocolates and Chocolate sweets and as such it has the exclusive right to use the trade mark. However, when the matter was listed for hearing Cadbury submitted that they were taking necessary steps for withdrawing the aforesaid registrations and therefore would not be arguing the matter on merits. ITC on the other hand submitted that that the matter may be heard and decided on merits as there was an injunction order operating against them.
In view of the facts and circumstances of the case IPAB was of the view that “Just the registration alone will not help the respondent (Cadbury) to prove their use. The impugned trademarks are liable to be removed on the ground of non-use. In view of the foregoing observations, the three rectification applications are allowed with a direction to the Registrar of Trade Marks to remove the trade mark.”
Bisleri barred from selling Maaza Mango Drink locally in India
In this case in the year 1993 Bisleri International had vide an agreement assigned the trade mark MAAZA including formulation rights, know-how, intellectual property rights, goodwill etc. to Coca Cola.

In 2008, Coca Cola filed an application for registration of the trade mark MAAZA in Turkey subsequently Bisleri sent a legal notice repudiating the said agreement. In view thereof, Coca Cola claimed permanent injunction and damages for infringement of trade mark and passing off.
Thereafter, the Division Bench of Delhi High Court on January 24, 2013 disposed off an appeal filed by Bisleri International thereby barring them from using the trademark Maaza to market its products in India. The Court further observed that Coca Cola would suffer irreparable loss and injury and accordingly granted interim injunction to Coca Cola Company.
Trademark Victory of some of The Well Known Marks- Amul, Bloomberg, Dr. Reddy’s, Kit Kat
Amul on Infringer MUL- Kaira District C-operative Milk Producres’ Union Limited who had been continuously and extensively selling milk, milk products, dairy products, food products under the mark AMUL since the year 1955 filed an appeal with Intellectual Property Appellate Board (IPAB) against impugned order passed by Registrar of Trademarks, Kolkata allowing trademark application no. 1281174 in class 29 filed by Ichamati Co-operative Milk Producers’ Union Limited for registration of the mark IMUL.

AMUL contended that it had been using its mark since the year 1946 and the same was duly registered in class 29 in early 1960. Moreover the sales turnover of the products sold under the trademark AMUL is in seceral crores of rupees. It alleged that IMUL is a label mark which is deceptively similar to its registered trademark and would definitely cause deception and confusion in the trade and public.
The IPAB while setting aside the impugned order passed by the Registrar of Trademarks observed that the marks AMUL and IMUL are deceptively as well as phonetically similar, thereby the possibility of confusion was certain.
Trademark Dilution Protection Widened: Delhi HC’s Order on BLOOMBERG- The present case was filed in the Delhi High Court seeking an injunction to restrain the defendants Prafulla Saklecha & Ors. from using the mark BLOOMBERG as a part of their corporate name. The plaintiff, Bloomberg Finance LP has been successfully running a 24/7 financial news channel since 2008 in India, and claims to be using the mark BLOOMBERG in India since 1996.
BLOOMBERG contended that the plaintiffs contended that the use of the mark by defendants constituted infringement and passing-off, thereby diluting the brand BLOOMBERG. The defendants were using the word BLOOMBERG in their company’s names, operating in the fields of construction and realty, food, entertainment, etc.
The Defendants on the other hand argued that the present dispute was essentially about the adoption of the mark BLOOMBERG as part of the trade name and Section 29(5) of the TM Act, 1999 was exhaustive on the issue of corporate name (as held by Bombay High Court in Raymond Limited v. Raymond Pharmaceuticals Private Limited) and if the Plaintiff was not able to make out a case under Section 29(5) of TM Act, 1999 then it was not entitled to any interim injunction. Section 29(5) provides that a registered mark is infringed if a person uses such registered mark as part of his corporate name and is dealing in same goods or services in respect of which the trademark is registered.
After hearing both the sides, the Hon’ble Court ruled in favour of the plaintiff and granted injunction against the defendants from using the mark BLOOMBERG. The Court opined that the plaintiff’s mark was a well known mark in India and essential elements for dilution were prima facie established. The plaintiff’s mark which is distinctive and well known worldwide can be associated with the construction or real estate industry which can prima facie be detrimental to the Plaintiff’s distinctive character and lead to dilution.
Reddy(ies) not Ready to give up REDDY- In this case the mark REDDY was adopted by the plaintiff i.e. Dr. Reddy Laboratories in 1984, however, it was applied for registration in 2001 in various classes. The defendant in the present case i.e.Reddy Pharmaceuticals Limited was an agent of Dr. Reddy Laboratories for 10 years but this association was terminated on April 1, 2003. After the said termination, Reddy Pharmaceuticals Limited filed application for the trade mark REDDY on April 17, 2003.
In 2005, the said trade mark of Reddy Pharmaceuticals Limited was registered in respect of the goods ‘medicinal and pharmaceutical preparations’ even when the said mark was filed subsequent to the mark REDDY filed by Dr. Reddy Laboratories.
In view of the facts and circumstances of the case at hand, the IPAB observed that Dr. Reddy’s Laboratories applied for the trade mark REDDY in 2001 whereas Reddy Pharmaceuticals Limited’s application for the trade mark REDDY was filed in 2003 i.e. subsequent to Dr. Reddy’s Laboratories’ application. Thus, the Trade Mark Registry made an error by registering the trade mark of Reddy Pharmaceutical Limited’s which was filed subsequently.
The IPAB held that Reddy Pharmaceuticals Limited had adopted the impugned mark to deceive the public and it was not a bon fide user. Thus, it could not take support of honest or concurrent use under Section 12 of the Trademarks Act.Sweet Victory for Kit Kat– This trademark dispute involved Societe des Produits Nestle S.A i.e. Nestle and Kit Kat Food Products headquartered in Kolkata.

In the year 1987, Nestle filed two trademark application in India for its KIT KAT Trademark however they were opposed by Kit Kat Food Products. Thereafter, in the year 1991 Kit Kat Food Products filed application for the mark Kit Kat for their various products. On being advertised the said marks were opposed by Nestle and were subsequently rejected by the Registry.
When the matter later on came up before the Intellectual Property Appellate Board (IPAB), it noted that Nestle had been using the mark KIT KAT outside India since the year 1935 and the impugned marks of Nestle were filed in India in the year 1987. It also noted that Nestle through evidence had established its use in India since November 01, 1987 and on the other hand Kit Kat Food Products had been using the mark KIT KAT since 1991 only.
In view thereof, the IPAB ruled that Nestle was the prior user of the mark KIT KAT and that the public will identify KIT KAT with Nestle and not Kit Kat Food Products.
INDUSTRIAL DESIGNS
Delhi High Court interprets “publication” in Industrial Design – Reckitt Benkiser India Ltd. v. Wyeth Ltd. (March 15, 2013)
The various provisions under the Design Act ensure that the designs which are not new or original are not registered under the Act. One of such safeguards enumerated under the Act is that a registered design can be canceled under Section 19 of the Act if such registered design has been published in India or in any other country prior to its date of registration. The legal interpretation as to what amounts to “prior publication” under the Act was interpreted in the judgment delivered by the Hon,ble Delhi High Court in the case of Reckitt Benckiser India Ltd. v. Wyeth Ltd.
The case at hand dealt with the design of S-shaped spatula used for depilatory purposes marketed by Reckitt Benckiser (India) Limited along with its hair- removal cream named “Veet” in India since 2004. Reckitt had obtained the design registration in respect of the S-shaped spatula vide registration No. 193988 dated December 5, 2003 in Class 99-00. Reckitt alleged that Wyeth Limited that has been selling the hair removal product under the brand name “Anne French” contains a spatula which infringes Reckitt’s copyright in its registered design. Reckitt approached the Court to seek an ad interim injunction against the Wyeth, restraining it from manufacturing, selling etc. the spatula with its hair removal product on account of its virtual identity with the design of Reckitt’s spatula.
This case for the first time came before Learned Single Judge of Delhi High Court on January 08, 2007. The Learned Judge after hearing the matter issued notice to Wyeth and passed an ex parte order wherein it was directed that till the next date of hearing, Wyeth, along with its representatives, agents etc. would be restrained from manufacturing/selling or offering for sale its products with a spatula which in any manner infringes the registered design No. 193988 of Reckitt.
Thereafter, Wyeth appeared before the Court and filed its application under Order 39 Rule 4 CPC seeking, inter alia, vacation of the aforesaid ex parte injunction passed by the court. Wyeth in its contentions before the Hon’ble Court mainly contended that Registration of impugned design is liable to be canceled under the provisions of Section 19 (1) (b) of the Act as the design had been published in other countries prior to the date of its registration in India and that Reckitt while obtaining design registration in India had concealed the fact that the design was prior registered in foreign jurisdictions like U.S.A., UK and Australia.

Reckitt retaliated stating that The Design Act says that prior publication either in India or abroad is a ground for cancellation of a registered design but only the prior registration abroad would not amount to cancellation of a registered design. It also contended that as per the Act a design which is new or original can be registered. Emphasizing on the word ‘or’ Reckitt submitted that even if the design may not have been new, it was definitely original as it originated from the author of such design.
The learned Single Judge vacated earlier issued ex parte injunction vide its order dated 7 August, 2009 stating that Wyeth has been able to show, prima facie, that the design had been disclosed to the public in UK, USA and Australia by publication in tangible form and that that there was enough evidence on record to show that the design had been published prior to the date of registration in India. While comparing the two designs, he held that any layman cannot conclusively say that one is an imitation of the other design, however the learned single Judge also put up a caveat that this opinion of his is only a prima facie opinion.
Aggrieved by the aforesaid decision of Learned Single Judge Reckitt filed an appeal against the said order before the Division Bench of Delhi High Court. The Division Bench of the Delhi High Court comprising of Hon’ble Justices Sanjay Kishen Kaul and Valmiki J. Mehta while agreeing with the Single Judge’s decision in not granting injunction to Reckitt on the basis of prior publication/existence in public domain through foreign registration referred the matter to a Larger Bench for considering the consequences of publication of the impugned design overseas which is not kept secret after registration.
The Hon’ble Court observed that Section 19(1)(a) of the Act has to be interpreted literally, and only a previously registered Indian design can be a ground for cancellation of subsequently registered design in India, and a foreign registered design cannot under Section 19(1)(a) be a ground for seeking cancellation of a design registered in India. The Court also held that once a foreign registered design is registered in India within six months of the date of application made in the convention country abroad, it becomes a design registered in India with an earlier priority date. Therefore, this registration can be a ground for cancellation of a design registered in India having a subsequent priority date.
On the issue of prior publication the Court held that according to Section 4(b) of the Act, it is not mere publication but publication “in a tangible form” or “by use” or “in any other way” which constitutes a ground for cancellation under Section 19 of the Act. In order to address the issue determining the extent there should be publication to fulfill the “tangible form” or “by use or any other form” requirement, the Court laid down the doctrine of “Sufficient Visual Clarity” According to this doctrine, in order to satisfy the conditions of publication “in a tangible form” or “in any other way” as prescribed under Section 4(b) the visual impact of the publication should be similar to the design as it visually appears on a physical object. The Court further observed that what amounts to publication is a question of fact and has to be decided on a case to case basis.
With respect to documents existing in the record of Registrar of Designs in a convention country, the Court held that design existing in the records of Registrar of Designs in a convention country abroad which are open to public inspection may or may not result in prior publication and that the main criterion which is to be considered in such cases is whether such document pass the test laid down in doctrine of “Sufficient Visual Clarity”.
After dealing with the above mentioned issues, the Full Bench of Delhi High Court upheld the decision of Division Bench as well as the Single Judge as to existence of prior publication of the impugned design of Reckitt’s spatula as it was found in actual use prior to its registration in India. The Court directed both parties to appear before the Single Judge for further proceeding.
COPYRIGHT
Copyright Activist, Aaron Swartz Commits Suicide

Aaron Swartz, a 26 year old activist who was heavily involved in copyright policy issues and issues surrounding technology freedom committed suicide on January 11, 2013. At the age of 14, Aaron had co-authored the ‘RSS 1.0’ specification. He was also part of the original Creative Commons technical team. He later also became a co-creator of the popular social news site Reddit.
He saw no sense in the American public having to pay for access to cases which were in the public domain and he strongly believed in making information more available to the public. Though there is no direct evidence for the reasons for his suicide a casual link has always been drawn considering the circumstances in which he committed suicide. The trend has been to somewhere blame the prosecutors of the US federal government for his suicide. He was arrested for charges of illegally downloading apparently 4.5 million academic papers from MIT and JSTOR resulting in possible jail time of over 50 years and 4 million dollars in fines. JSTOR apparently had dropped the charges, but the US government continued the case and racked up a total of 14 counts of felony against him.
Delhi HC rejects the ‘Hot News’ Doctrine in Cricket Match Alerts
The applicability of the ‘Hot News’ doctrine was rejected recently in a landmark ruling delivered by Justice Bhat of the Delhi HC. In 2012, by an Agreement, Board of Cricket Control of India granted exclusive broadcasting rights to Star TV to disseminate the information/content emanating from the cricket matches which included the right to record, reproduce, broadcast etc. Sometime later, Cricbuzz, Idea Cellular and ONMOBILE started SMS services providing contemporaneous ball-by ball coverage of live cricket matches. The ‘mobile distribution’ rights were the bone of contention The Single Judge of the Delhi HC rejected the defendants’ contentions and the ad-interim injunction restraining the defendants from disseminating contemporaneous match information in the form of ball-by-ball update/match alerts for a premium was granted. The Division Bench of the Delhi HC overturned the decision in the present judgment.
The Court discussed the Hot News doctrine and its application in India. The doctrine has been narrowed to injunct time sensitive news where both parties are “direct competitors” and not merely where the plaintiff’s primary service or product is not hot news dissemination, but match organisation or broadcasting of those events. This critical aspect of ‘Hot News’ was absent in the present case, as neither Star, nor BCCI engaged themselves primarily in match news dissemination through SMS. Thus, the respondents failed to show to the court, how it had proprietary rights over the facts and information it sought to protect – even for a limited duration. Furthermore, the Appellants had argued that their right to freedom of speech and expression and their right to freedom of trade under Article 19 (1) (a) and (g) could not be interfered with in the absence of any law.
Delhi University/Photocopy Case: Highlighting Developments
Background
A brief trajectory of the Delhi University-Photocopy Case law suit:
- Leading publishers sued Delhi University (DU) and its photocopier (Rameshwari) for copyright infringement in August 2012.
- An injunction restraining DU and the photocopier was granted in October 2012 prohibiting course packs i.e compilations/photocopies from books.
- Delhi University appealed against the order in November 2012. The appellate court asked them to file a review petition before the single judge. However they opted to file an Order 39 Rule 4 application for vacating the restraining order.
Student’s Intervention Petition
An intervention petition was filed on behalf of the “Association of Students for Equitable Access to Knowledge” on March 1, 2013. The publishers’ naturally objected to the student intervention and argued that even if intervention is admitted, it should be limited to points of law. The Delhi High Court however disagreed and held that the student association would be admitted as a full fledged party to the law suit, with the ability to file a written statement and lead evidence etc.
Author’s Letter requesting Publisher’s to Withdraw the copyright suit
The case took an interesting turn when the very authors and academicians whose larger interests the Publishers (Oxford University Press, Cambridge University Press and Taylor & Francis) are seeking to represent in the copyright suit, on the contrary expressed their solidarity with the students by sending a letter to the said Publishers requesting them to withdraw the suit.
The letter has been sent by a group of as many as 309 writers and academicians from all over the world to the publishing houses of which 33 signatories to the letter had been specifically mentioned by the publishing houses to have contributed to the work that forms the subject-matter of the dispute.

The authors have expressed their opinion in the letter that the ‘course-packs’ prepared by D.U. fall firmly within the ambit of statutory exceptions to copyright infringement (‘fair use’), being covered under Sections 52(1)(a) and 52(1)(n) of the Indian Copyright Act, 1957. The letter also rightly rejected the fallacious assumption that a ban on photocopying would necessarily result in increased book sales. It highlighted the fact that in view of the exorbitant prices charged by publishers, students could not afford to buy these text books anyway.
Academicians “Speak” Out
Another significant milestone in the unfortunate copyright case was when the court permitted SPEAK (“Society for Promoting Educational Access and Knowledge”), an association of academics and authors to intervene on April 20, 2013. The SPEAK petition requests for an impleadment on the ground that the law suit filed by the publishers effectively targets all educational institutions that create and distribute course packs.
To allow the plaintiffs to commercially exploit the labour of salaried academics, and to reap the benefits of having their works prescribed as part of the syllabus, while simultaneously restricting the right of the University to make available copies in the course of instruction will do serious injustice to the balance in the Copyright Act between the rights of owners of copyright and the right of the Applicants and students to access educational/instructional material.
Solution to the Photocopy Row – IRRO’S licensing policy?
The copyright battle has reached the next stage, with Indian Reprographic Rights Organisation (IRRO) claiming that there is a legal way to photocopy course material which will protect students’ interest without violating Indian Copyright Act.
Well, all in all, one can only say for sure that no matter what the court finally decided in this case, this is going to be a raging issue for quite a while to come, given it has so much at stake, both from the students’ and academicians’ side as well as the publishers. In what started off as a brief spark in the Indian copyright scenario is fast being stoked into a raging fire all over the country and beyond.
ANNUAL REPORTS
Intellectual Property Appellate Board’s Annual Report (2012-2013)
Some of the noteworthy points as covered in the Indian Intellectual Property Appellate Board’s Annual Report are presented below:
- There has been a significant increase in the number of cases filed before IPAB. Despite of lack of funds and human resources the Board was successful in maintaining its performance.
- Various cases which were dealt and decided by the board and were thereafter also followed by the Judiciary in pronouncing its judgment were:
- Spice Mobiles Ltd. v. Shri Somasundaram Ramkumar (Dual Sim Case)
- Tata Chemicals Limited v. Hindustan Unilever Limited (Pure It Case)
- Bayer Corporation v. Union of India (Appeal against the first ever order of Compulsory License passed by the Controller General)
- Sankalp Rehabilitation Trust v. F. Hoffman- La Roche AG (Pegasys)
- Hindustan Unilever Limited v. M/s Three-N-Products (P) Ltd. (Ayur)
- Relaxo Footwears Limited v. Bata India Limited
- The ambiguity in Divisional Applications was resolved and it was held that there would be no divisional application if there is no plurality of invention
- The efforts made by the Board in the year 2012-13 are highlighted below:
- The Board’s website throughout the year reflected the notifications and circulars issued by it, thereby enabling the stakeholders to easily access the same;
- For smooth functioning of the system, the Board’s initiative to amend the rules and procedures involved in filing of Applications received a green signal from the Central Government and the same is expected to be finalized soon.
- In order to strengthen its system, the IPAB submitted a Detailed Project Report to the Central Government (as per the Plan Scheme of the Government -12th Five Year Plan)
- Hurdles faces by IPAB as highlighted in the report were:
- Shortage of human resources;
- Lack of proper infrastructure;
- Paucity of funds;
- Unconstitutional appointment procedure.
The Report also acknowledged the co-operation and contribution of the Bar and aspires that in the years to come the Board would be perceived as a model- Tribunal.
Indian Intellectual Property Office’s Annual Report (2011-2012)
The PATENTS
The number of applications filed for grant of patents in 2011-2012 was 43197 while in 2010-11 the figure was 39400, showing an increase of about 9.64%.
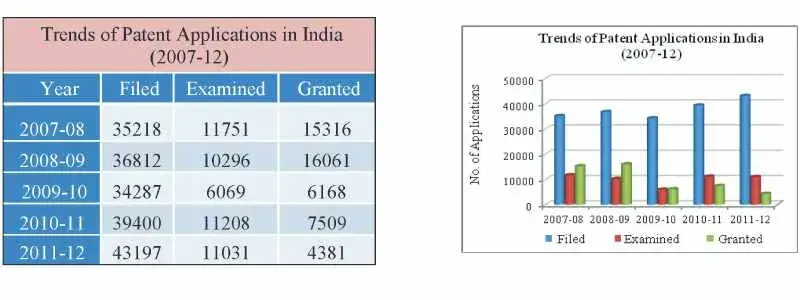
Fig 1: Table and Graph depicting trends of patent applications filed, examined and registered in the last five years (2007-12)
The Report stated that the maximum number of patent applications was filed through Patent Cooperation Treaty (PCT) route. The United States of America (9137) led with the maximum number of applications followed by Japan (3980), Germany (3405), and Netherlands (1642) etc. The other top filers are as follows:
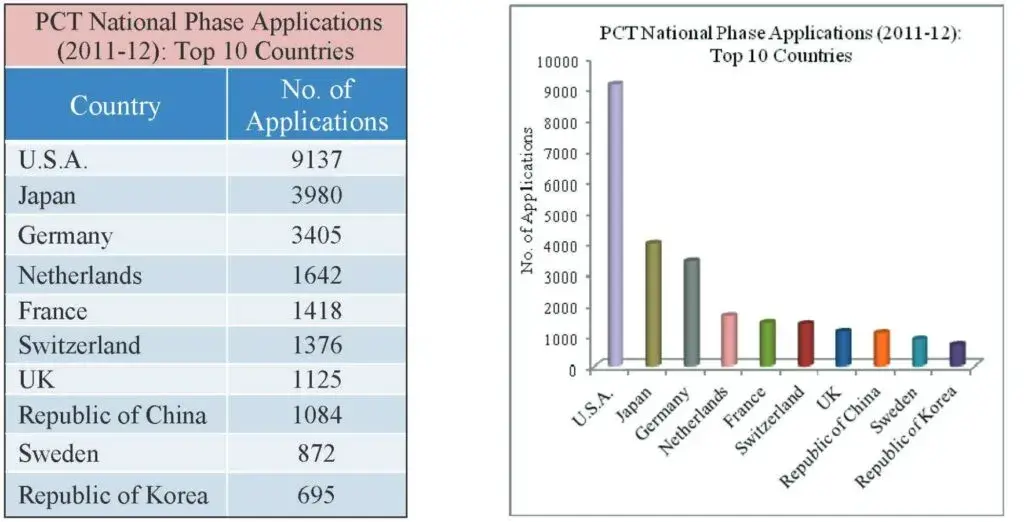
Fig 2: Table and Graph of Top 10 Countries for filing PCT National Phase Applications
The top Indian and foreign applicant as well as the patentee were Council for Scientific and Industrial Research (CSIR) Qualcomm Inc. respectively.
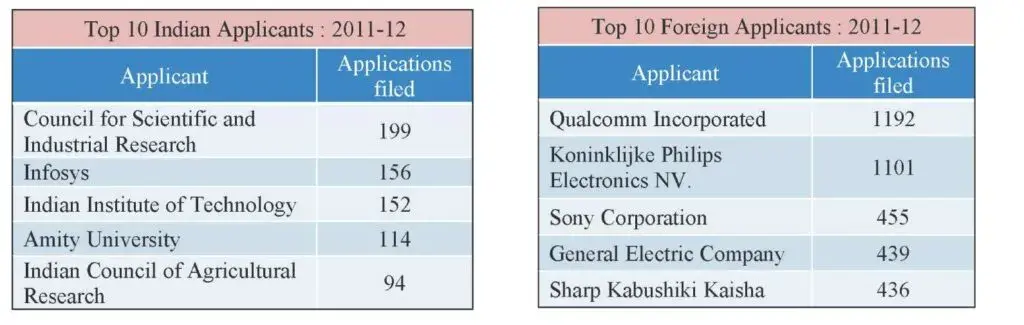
Fig 5: Top 5 Indian Patentees
Fig 6: Top 5 Foreign Patentees
DESIGNS
In the year 2011-12, 8373 design applications were filed, which showed an increase of about 10.33% as compared to last year.
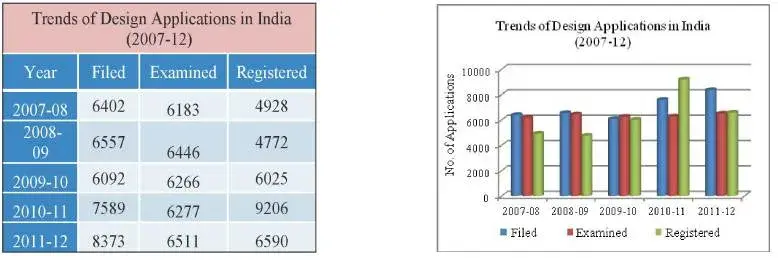
Fig 7: Table and Graph depicting trends of design applications filed, examined and registered in the last five years (2007-12)
In the case of applications originating from foreign countries, with 811 applications the U.S.A was the top filer followed by Japan (570), Germany (373), Switzerland (244) and U.K (244).
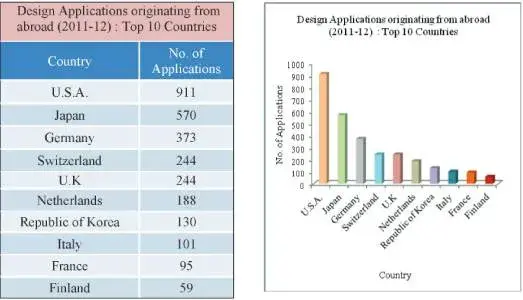
Fig 8: Table and Graph depicting the top foreign countries from which maximum design applications were filed in India – 2012
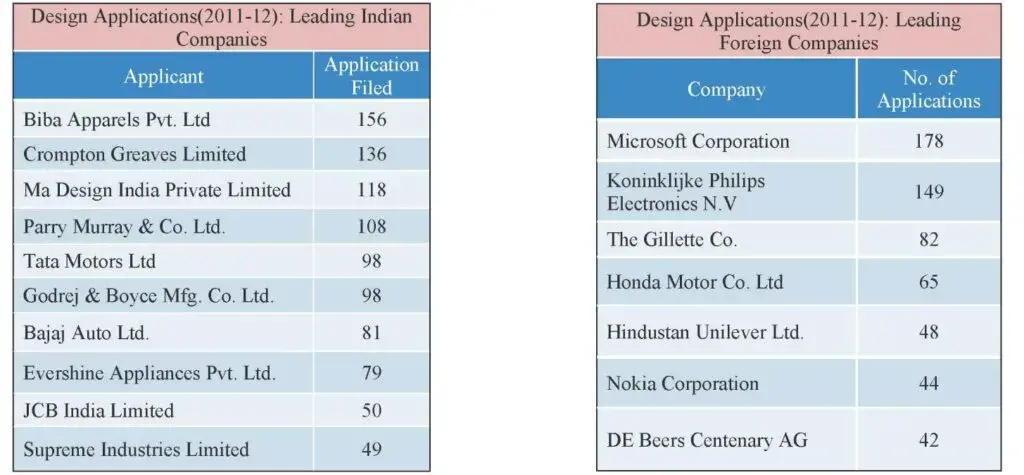
The leading Foreign Companies were Microsoft Corporation (178) Koninklijke Philips Electronics N.V (149), The Gillette Co. (8.2) and Honda Motor Co. Ltd (65). Similarly, leading Indian companies were Biba Apparels Pvt. Ltd (Filed 156, Regd.225), Crompton Greaves Limited (Filed 136, Regd. 151), Ma Design India Private Ltd. (118 Regd. 69) Parry Murray & Co. Ltd. (Filed 108, Regd. 90) and Tata Motors Ltd (Filed 98, Regd. 42). Other top filers are mentioned in the table below.
TRADEMARKS
In the year 2011-12, 183588 trademark applications were filed, showing an increase of about 2.38% over the previous year. However, there was a decrease in the number of applications examined and registered as compared to the previous year. The Annual Report states that this was mainly due to the commencement of the new system of centralised examination of applications at the Mumbai Trademark office. The trend of the last five years is given below.
The filing trend of applications indicates that the number of applications filed for this year (2011-12) was more than in the previous year. In comparative terms, there was an increase of 4271 number of applications over the previous year.
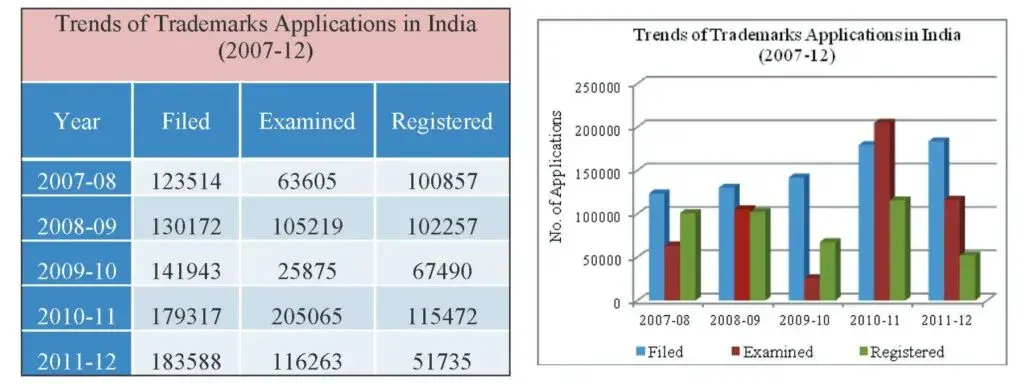
Fig 11: Table and Graph depicting trends of trademarks applications filed, examined and registered in the last five years (2007-12)
Altogether 1,21 ,006 device marks type of application were filed which is about 65.91% of total filing and similarly about 62,205 word-mark type of trademarks applications were filed which is 33.88% of total filing.
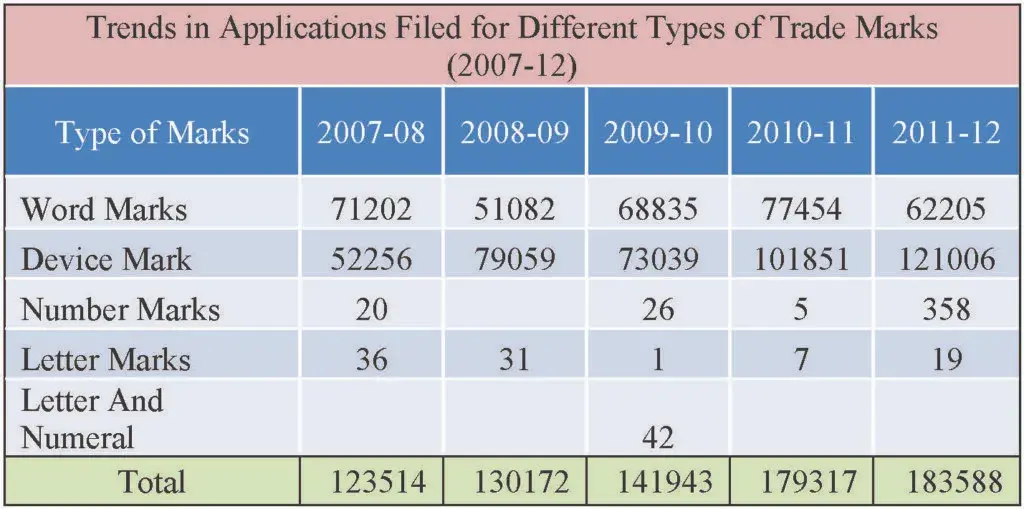
Fig 12: Types of trademarks filed in the last five years (2007-12)
As in the previous year, for this year also the largest number of applications was filed in respect of goods in Class 5 (Pharmaceuticals, Veterinary and Sanitary Substances etc).
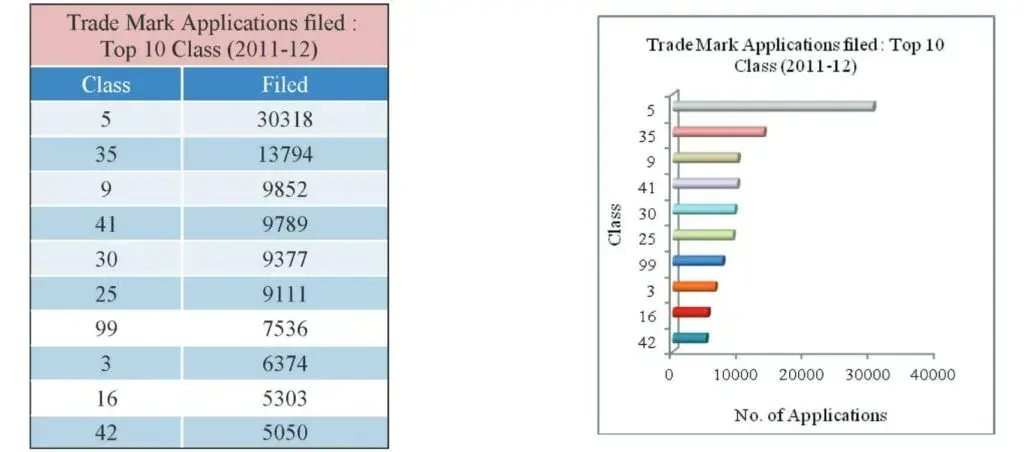
Fig 13: Table and Graph depicting the top classes in which maximum trademark applications were filed (2012)
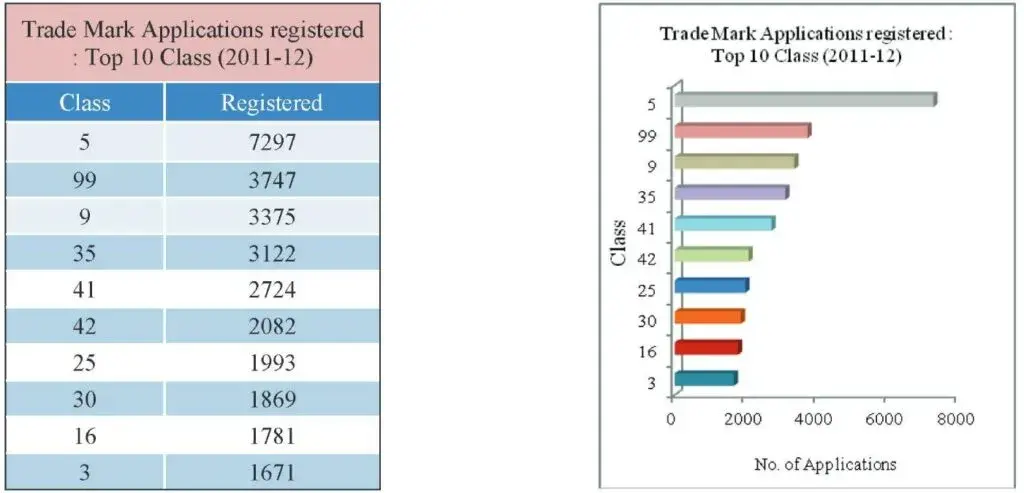
Fig 14: Table and Graph depicting the top classes in which maximum trademark applications were registered (2012)
GEOGRAPHICAL INDICATIONS
The Registry started receiving GI applications for Registration from 15th September 2003. From September 2003 to March 31, 2012, the Registry has received a total number of 380 GI Applications.
In 2011-12, 148 GI Applications were filed and the Registry has received 145 GI Authorised User Applications during the financial year. A total of 23 Geographical Indications were registered during the year 2011-12
A total of 23 GI registration certificates and 116 GI Authorised User certificates were issued by the Registry in 2011-12.
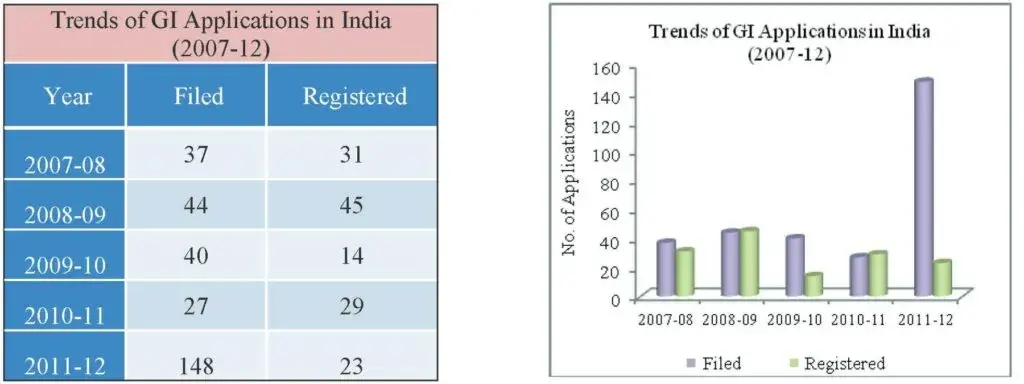
Fig 15: Table and Graph depicting the trend in GI applications filed and registered over the last five years is provided below.
UPDATES FROM THE INDIAN INTELLECTUAL PROPERTY OFFICE (IPO), DEPARTMENT OF INDUSTRIAL POLICY AND PROMOTION (DIPP), COPYRIGHT OFFICE
- The Indian Intellectual Property Office (IPO) notified the applicants/agents to verify the Cash Book Receipts (CBRs) issued by IPO to ensure that payments intended for certain documents have actually been attributed to such documents. All the Agents and Applicants were also directed to bring any error in the CBR to the notice of the Patent Office immediately. (February 1, 2013).
- The Office of Controller General of Patents, Designs and Trademarks (CGPDTM) issued a notification directing all Patentees and Licensees (whether exclusive or otherwise) to furnish information and statement regarding the commercial working of patented invention in India (in compliance with Section 146 and Rule 131 of the Patent Act). Vide the said notification; the Patent Office also appealed to all the applicants and agent to use, as far as possible, the IPO’s e-filing services for filing Form 27. (February 12, 2013).
- CGPDTM made available to the public complete details of all the pending and registered Geographical Indications Applications. All the documents with respect to the GI applications including copy of Application Forms, Statement of Case, Examination Report, E-Register of Registered GI applications and other supporting documents and necessary details were being made available by the Registrar. (February 14, 2013).
- The Copyright Division, Department of Higher Education, Ministry of Human Resource Development notified the Copyright Rules, 2013. The introduction of new provisions under the Copyright (Amendment) Act, 2012, which came into force on June 21, 2012 led to amendments in the Copyright Rules, 1958. Among other amendments were the considerable enhancement in the fee for registration of copyright for various works and fee for licenses to be issued by register of Copyrights. The minimum fee for registration was increased from Rs. 50/- per work to Rs. 500/- per work; while the maximum fee for the same has been enhanced from Rs. 600/- per work to Rs. 5,000/-. Similarly the fee for licenses was increased from Rs. 200/- to Rs. 2000/- per work and the maximum fee was increased from Rs. 400/- to Rs. 40,000/. (March 19, 2013).
- India joins Madrid Protocol System – Commerce and Industry Minister Anand Sharma submitted India’s instrument of accession to the Madrid Protocol for the International Registration of Marks at WIPO. With this treaty India became the 14th of the G-20 economies to accede to the Madrid Protocol. (April 9, 2013).
- The Ministry of Commerce and Industry released a notice in the Official Gazette of India pertaining to the draft of amendment of Patent Rules 2003, which the Central Government proposes to make. The major amendments proposed in this regard were: a) The official fee payable in respect of the grant of the patents and applications therefore, and in respect of certain other matters for which fees are required to be payable under the Act to be increased by upto 100 percent, b) 10 per cent additional surcharge to be levied on the fees for different proceedings under the Patents Act 1970 (as amended) when the application for patent and other documents are filed in physical mode namely, in hard copy format, c) A new Form 7 (A) specified for filing Pre-Grant Opposition and d) The fee for various matters in respect of which the cost is to be awarded by the Controller to be increased in certain cases with a proviso that that 10 per cent additional surcharge shall be levied on the scale of the costs when the documents are filed in physical mode, namely, in hard copy format. (May 6, 2013).
- A dynamic utility grid made available by the Patent Office grid to enable the applicants and patent agents to view the month of Request for Examination, for which the Office Action is currently being issued by the various branches of the Patent Office. The dynamic grid displays the information about the month and year of Request for Examination (Form 18) that are being examined and FER being issued in each examination group i.e. Electrical/Electronics, Mechanical, Chemistry and Biotechnology. (June 17, 2013)
- The Office of Controller General of Patents made available all the information regarding “Statement of Commercial Working of Patents” (in accordance with section 146(3), read with rule 131(3) of the Patents Act) as received from the patentees for the year 2012. The portal made available by the Patent Office enabled users to retrieve information using criteria such as Patent No., Application No., and Name of Patentee across all four locations of the Patent Office. (June 25, 2013).
- The Patent Office notified that the “Comprehensive e-filing services for Patents” would also enable the applicants to submit reply to the Office Actions, including reply to the first and second objection report, electronically. The notice however further stated that applicants shall be required to submit the Original copies of documents such as Power of Attorney and Certified Copy of Priority Documents with the Patent Office. (July 7, 2013).
- The Intellectual Property Office (IPO) notified that appropriate reply to the objections raised in the Examination Reports, (with respect to Trademarks), were not being submitted by the applicants, advocates/ registered trademark agents and stakeholders. The CGPDTM advised the trademark holders, advocates/registered trade mark agents and stakeholders to submit a proper reply to the examination report under the heading “REPLY TO EXAMINATION REPORT” which would enable the Examiner to dispose off the case on merit under the PARM Module. The Notice also underlined few guidelines with respect to filing of evidence. (September 24, 2013).
- The Intellectual Property Office (IPO) launched Dynamic Patent Utilities which would enable the patent holders to search for patents and also ascertain its current status. The Dynamic Patent Utilities module displays information in comprehensive manner on Patents – that have expired, which have ceased to have effect by reason of failure to pay the renewal fee and according to their Number, Title & Technical / Scientific field, which have expired or have ceased to have effect by reason of failure to pay the renewal fee. (September 26, 2013)
- The Indian Patent Office was empowered as an International Searching Authority and International Preliminary Examining Authority. The agreement between the Indian Patent Office and the International Bureau of the World Intellectual Property Organization in relation to the functioning of the Indian Patent Office as an International Searching Authority (ISA) and International Preliminary Examining Authority (IPEA) under the Patent Cooperation Treaty (PCT) has come into force (from October 15, 2013) and shall subsist until 2017 unless terminated in accordance with the agreement. (October 15, 2013)
- Amendments in PatentRules: Following designation of India as the International Searching Authority and International Preliminary Examining Authority, the Central Government amended Patent Rules in exercise of the powers conferred by section 159 of the Patents Act, 1970 (39 of 1970). The prominent amendments were made in Rule 9 (Filing of documents and copies, etc.), Rule 18 (Appropriate office in relation to International applications) and Rule 19 (International applications filed with appropriate office as receiving office). October 15, 2013.
- On the lines of Dynamic Patent Utility tool, the Trademark Registry launched the Dynamic Trademark Utility. Through this tool public can generate a report displaying the number of applications examined, number of show- cause hearings issued, number of trademarks published in the Journal, number of trademarks registered, number of trademark applications disposed or number of notices issued by simply selecting the desired detail and feeding the relevant month and year for the same. (November 1, 2013).
- In a progressive step towards digitization and up gradation of Trademark Registry, the Intellectual Property Office (IPO) notified that the replies to office actions/ examination reports can be filed online. The replies to office action along with the supporting documents can be uploaded online through the comprehensive e-filing services of trademarks module. (November 07, 2013)
- The Trademark Registry issued a public notice notifying initiation of a special drive for disposal of requests filed for recordal of post-registration changes in the registered trademarks. The said drive will take place during the months of December, 2013 and January, 2014. The Registry notified that it shall be taking Requests filed on Forms TM- 17, 19, 20, 23, 24, 33, 34, 35, 36, 38, 42, 43 or 50 for disposal. It had also notified that all the requests for recordal of post registration changes would be disposed in sequential order of changes. The documents that an Applicant/Agent were to bring for post registration changes including Statement of case indicating sequence of changes, Authentic copy of assignment deed and Power of Attorney and Affidavit indicating that no litigation is pending with respect to the assignment/transmission mentioned in the request; were also enlisted in the notification. (November 20, 2013).
- Owing to poor response of the Agents/Applicants, the CGPDTM had issued another Public Notice requesting the Applicants/agents to participate in the drive so that the post- registration changes could be disposed off. (December 13, 2013).
- In continuation of the aforesaid, the Registry issued another notification in which various officers of the Trademark Registry who are designated under special drive to dispose of requests for recordal of post registration changes in the registered Trademarks were allocated cases to for the month of January 2014. (December 24, 2013).
- CGPDTM by virtue of the power under Section 8(1) of the Trademarks Act, published classification of goods and services for the purpose of registration of Trademarks in India. The classification as published consisted of Introduction, Class Headings, List of Classes with Explanatory notes, Alphabetical List of Goods, Alphabetical List of Services etc. (November 29, 2013).
- IPO notified that it would be issuing Renewal Certificates of patents with QR Codes printed on it. It was also notified that such certificates shall also be made available to the applicant/agent on record immediately by email. The IPO stated that it would be extending the service to SMS in due course. The Patent Office has further notified that although as of now this service is offered only for renewal certificates, it intends to extend the same to various stages of processing of patent applications as well. (December 11, 2013).
- CGPDTM issued public notice stating that the Document Receiving Counters situated in front of Trademark Registry premises would be equipped with Computer Monitors, thereby facilitating scrutinization and verification with respect to the data entered in the electronic system at the receiving counters. (December 23, 2013)
S. S. Rana & Co.’s contribution in IPR sensitization
S.S. Rana and FICCI’ Law Book on Publishing Industry: In order to capture new developments in copyright laws and to provide publishers in India a ready reference of all legal aspects pertaining to publishing industry, S.S. Rana & Co. in association with Federation of Indian Chambers of Commerce and Industry (FICCI) authored and came out with Publishers’ Law Book. The law book was released by Mr. Younhshuk Chi, President International Publishers Association and Chairman Elsevier U.S.A at the seminar on Legal Aspects of the Book Publishing Industry that was organized by FICCI. Mr. Vikrant Rana, Managing Partner S.S. Rana & Co. addressed the publishers and industry experts who attended the seminar and deliberated on the legal framework and issues affecting the publishing industry as well as on other topics including copyright laws that were covered in the book.
The book brings together important developments in the legal and regulatory framework in Indian publishing industry and covers disparate topics such as Current Trends in the Publishing Industry, Emergence of Digital Era, Statistical Analysis of the Print and Publishing Industry in India and IPR Management. The book also throws light on technological developments in production, consumption and distribution that is currently being witnessed by the Publishers in India and abroad.
A copy of the book can also be accessed from the following link: https://ficci.in/public/api/press_release_details/4702
Script IT- Essay Writing Competition on the occasion of World IP Day (April 26, 2013) and Creatio- A design competition on the occasion of World Industrial Design Day (June 29, 2013) In order to give young students an opportunity to understand the role played by intellectual property rights in protecting the work and creation of human mind and intellect & to raise awareness on IP Rights India, Vidya Darshan Rana Charitable Trust in collaboration with S.S. Rana & Co. had organized All India Essay Writing Competition on IP relevant theme. The competition aimed to provide a platform to the students to share their views and would enable them as well as their college in getting national recognition.

“Creatio” was organized with the aim to raise Intellectual Property awareness in India and help the grassroots innovators, designers, artisans and creators to realize their true potential by providing a national platform to them. The competition was recognized and acknowledged by International Society for Complexity, Information, and Design (ICSID).
Participation of Mr. Vikrant Rana, Managing Partner, S.S. Rana & Co in seminars and conferences organized by government and non-government entities in 2013
- Delivered a lecture on “An overview of Trademarks-Trademark Litigation in India and abroad” in the lecture/seminar organized by TIFAC, Dept of Science & Technology – (WOS-C Scheme), in New Delhi on January 24, 2013.
- Delivered a lecture on “Trademark Searches” in the lecture/seminar organized by TIFAC, Dept of Science & Technology – (WOS-C Scheme), in New Delhi on January 24, 2013.
- Deliberated on the “Common Issues of Publishing Industry” in the “CEO SPEAK” session organized by FICCI in New Delhi on February 5, 2013
- Delivered lecture on “Industrial Designs” at the Delhi University on April 25, 2013
- Delivered presentation on “Importance of Trademark in Development of MSME and Perfumery Industry” in the seminar organized Intellectual Property Facilitation Center, Fragrance and Flavor Development Center, Kannauj in New Delhi on October 28, 2013
- Delivered Presentation on “Trademark and its importance in Marketing and Branding of Goods” in the seminar organized by Council of Science and Technology, UP in Lucknow on December 18, 2013.
- Authored a chapter on “Intellectual Property Management” for the book compiled by Madhya Pradesh Council of Science and Technology on Intellectual Property Rights.
Other contributions by the firm
- Ms. Rishu Srivastava, Patent Agent, deliberate on Patent Agent Examination system in India in the lecture/seminar organized by TIFAC, Dept of Science & Technology – (WOS-C Scheme), in New Delhi on January 30, 2013.
- Ms. Ankita Mohanty, Advocate, deliberated on the legal issues affecting the Small and Medium Enterprises, with the objective to educate the SMEs and increase their awareness on the intellectual property rights and its relevance for the SMEs, in the “7th SME Sammelan – Banking and Financing Solutions for Sustainable Growth” organized by ASSOCHAM in Dehradun on April 25, 2013.
- Mr. Rupin Chopra, Advocate, presented lecture on Intellectually Property Rights particularly trade marks and its importance thereto in relation to businesses in the 8th SME’s -“Sammelan”- Enhancing Capacity for Sustainable Growth organized by ASSOCHAM in Bhopal, Madhya Pradesh on June 5, 2013.
- Ms. Rishu Srivastava gave presentation on Patents and Industrial Design in the seminar organized by National Institute for Entrepreneurship and Small Business Development in Saharanpur, UP on December 11, 2013.
- Mr. Vikrant Rana and Ms. Rishu Srivastava submitted feedback to the Department of industrial Policy and Promotion in response to the notification dated May 6, 2013 issued by DIPP regarding draft of certain rules further to amend the Patents Rules 2003, which the Central Government proposes to make. (June 19, 2013)
- Ms. Ankita Mohanty’s, Ms. Hema Shekhawat’s and Ms. Shilpi Saurav Sharan’s, Associate Advocates, paper on “Book Exports, Piracy and Parallel Imports in India” was selected and published by FICCI. The paper was distributed amongst all the participants of the “PubliCon – 2013” conference organized by FICCI in New Delhi from September 10-11, 2013.
Our associates and research analysts have also written for several esteemed national and international journals
FACTOPEDIA
Our Annual Issue’s factopedia discusses some of the funnies patents ever…
- Shark Protector Suit – Patent Number: 4833729
A shark protector suit, it is a combined rubber suit and helmet to completely cover the body of the wearer, including a face mask for facial protection, and preferably having at least a partial lining of flotation material, such as foam sheet.
But how does it protects against sharks- well the suit and the helmet have a plurality of spikes extending outward therefrom to prevent a shark from clamping its jaws over the wearer.
So next time before going out for swimming you know what preparation you have to do.
- Scent Delivery System – Patent Number: US 6842218
Probably scent of any other entity, but of himself would have been so repelling for the inventor that it prompted him to invent “The scent delivery system” so that the user can pick his or her own smell.
This system employs a single conduit with a nasal interface at one end and a fan or canister of compressed air at the ether end. In between the two ends a scent injection device is positioned. The injection device injects scent into an air flow created by the fan or canister and creates scented air which is carried to the nose by the flow of air.
- Collars – Patent Number: US D504,984
If your favorite fashion brand is not sensitive enough to design matching accessories for your dog, no need to worry – the inventor here has the solution.
The Matching Collar for Dog and Owner – As per the inventor the collar’s design will enhance the necks of both the owner and the dog and that the matching collar for dog and owner is quite versatile and can also be enjoyed casually as well.
- Combined Clothes-Brush, Flask and Drinking Cup – Patent Number: 490964
This invention would prompt you to sing… “All I want is everything…everything at once”… for the object of the invention is to construct a combined clothes brush, liquor flask, and drinking cup, to provide the compact disposal of the parts.
So now you can store the drink, have it whenever you want and if need be can tidy yourself up.
- Portable Bar – Patent Number: 4681244
Ever heard of “Wherever you go, our network follows”… well now wherever you go, the mini bar (that too on your head) can follow you.
Here is the invention which is a a portable mix bar and comprises a hard hat having a plurality of drink containers and a pair of mixing chambers for mixing drinks mounted thereon wherein the flow of drinks to and from the mixing chambers is controlled by a plurality of valves.
Also provided is a plurality of tubes for carrying drinks from the drink containers to the valves and mixing chambers and between valves and mixing chambers and from the mixing chambers to the mouth of a user.
IP CONNECT WISHES ITS READER A VERY HAPPY AND PROSPEROUS NEW YEAR -2014
Like last year, the Indian intellectual property regime received more than called for attention due to the decisions of the judicial bodies for cases such as Novartis’s (Gleevec), Bayer (Compulsory License for Nexavar) and other revoked patents (GSK, Allergan etc.). Undeniably India’s image is portrayed as the “Anti-patent” nation, leaving multi-national pharma in a tussle – whether to ignore this emerging market of the world or to enter and then grapple with the enforcement issues, (more so because India’s IP policies may be emulated by other BRICS countries).
But is India actually an “Anti-patent” nation? Is the patentee not at fault for ignoring the statute? The provisions of the Indian Patent Act, explicitly lays down the conditions for the patentability, which governs all these proceedings. The onus indeed is on the applicant to formulate appropriate patent prosecution strategies such that not only the procedural and administrative requirements are complied with, but the patent can also be maintained in any litigation where validity of the patent is questioned.
If we look at the Novartis case howsoever strict or stringent the debatable Section 3(d) is, it is after all the statute that governs patent rights in India and is required to be complied with before the patent is granted. As also noted by the Supreme Court in the said case “…The aforementioned amendment in section 3(d) is one of the most crucial amendments that saw the Bill through Parliament … The amended portion of section 3(d) clearly sets up a second tier of qualifying standards for chemical substances/pharmaceutical products in order to leave the door open for true and genuine inventions but, at the same time, to check any attempt at repetitive patenting or extension of the patent term on spurious grounds …”
Now if we compare the Indian provisions with that of EU and US, it is the US laws on patenting that are most liberal and does not as such provide any list of inventions that are non patentable. The EPO, besides other restrictions, provides exceptions to patentability to plant or animal varieties or essentially biological processes for the production of plants or animals; this provision shall not apply to microbiological processes or the products thereof; and Methods for treatment of the human or animal body by surgery or therapy and diagnostic methods practised on the human or animal body; this provision shall not apply to products, in particular substances or compositions, for use in any of these methods. (EPC Article 53)
That the statutes of these countries itself do not provides for any similar provision like that of 3(d) itself explains why these patents are granted in other countries but not in India. Besides the patent rights are territorial and it should not be an issue as to why a particular patent is granted in one State and not in other. India has specifically framed the policies to inhibit ever greening and unfair competition and abuse of patent rights.
Patent prosecution in India is a resource intensive process, both in terms of money and time. It is therefore of utmost importance for the applicants to be aware of the nuances of the Patent Act. For strong patent enforcement the applicants must take care of the provisions of the Act during initial stages of prosecution itself so that, as and when the situation arises, it could be appropriately defended and enforced.
Quite literally liberal grant of patents would prove to be a major setback for generic drug manufacturers who can provide equally efficacious medicine at a very cheaper rate. Cases such as the one covered in the Newsletter would infact aid the innovator companies in properly analyzing their products before entering the patent regime in India.
But then all was not bad after all, India rejected BDR pharmaceutical’s application for Compulsory License too. In cases of Kibow Biotech and GlaxoSmithKline, the dispute were related to two patents of each entity, while one patent was revoked the other, in both cases, were upheld by IPAB, quite obviously the denied/revoked patents attracted a large media attention. There were major developments in the IP front as well; the most significant being long anticipated accession to Madrid Protocol. India also made legislative reforms and aligned its Intellectual Property regime with the global norms through the Trademark (Amendment) Bill, 2009 which led to the inclusion of a new chapter (Chapter IV A) in the Trademarks Act. India became the International Searching Authority. There were noteworthy administrative developments in the IP Office as well (as discussed above) and progressive steps were taken towards technological advancement, digitization, up gradation and modernization of Intellectual Property Office to enhance transparency in the public delivery system.
With this note IP Connect wishes all its readers a very happy, bright, cheerful and prosperous New Year 2014.
DISCLAIMER: The newsletter intends to provide general information only and should not be taken as legal advice or opinion related to specific situations. Every possible effort has been made to ensure accuracy of the information and statistical figure contained in the newsletter but the firm cannot be held responsible for any misrepresentation or inaccuracy.








